Impact Factor
Theranostics 2021; 11(19):9198-9216. doi:10.7150/thno.62455 This issue Cite
Research Paper
LOXL2-enriched small extracellular vesicles mediate hypoxia-induced premetastatic niche and indicates poor outcome of head and neck cancer
1. Department of Head and Neck Oncology, West China Hospital of Stomatology, State Key Laboratory of Oral Diseases, National Clinical Research Center for Oral Diseases, Sichuan University, Chengdu, 610041
2. Sichuan Key Laboratory of Radiation Oncology, Sichuan Cancer Hospital & Institute, Sichuan Cancer Center, School of Medicine, University of Electronic Science and Technology of China, Chengdu, 610041
Abstract
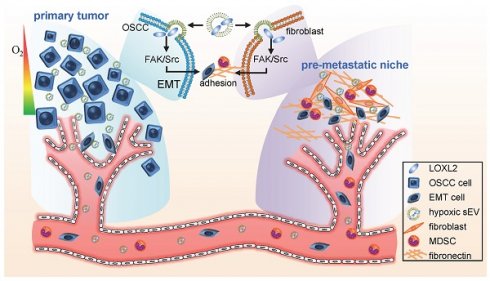
Small extracellular vesicles (sEVs) operate as a signaling platform due to their ability to carry functional molecular cargos. However, the role of sEVs in hypoxic tumor microenvironment-mediated premetastatic niche formation remains poorly understood.
Methods: Protein expression profile of sEVs derived from normoxic and hypoxic head and neck squamous cell carcinoma (HNSCC) cells were determined by Isobaric Tagging Technology for Relative Quantitation. In vitro invasion assay and in vivo colonization were performed to evaluate the role of sEV-delivering proteins.
Results: We identified lysyl oxidase like 2 (LOXL2) which had the highest fold increase in hypoxic sEVs compared with normoxic sEVs. Hypoxic cell-derived sEVs delivered high amounts of LOXL2 to non-hypoxic HNSCC cells to elicit epithelial-to-mesenchymal transition (EMT) and induce the invasion of the recipient cancer cells. Moreover, LOXL2-enriched sEVs were incorporated by distant fibroblasts and activate FAK/Src signaling in recipient fibroblasts. Increased production of fibronectin mediated by FAK/Src signaling recruited myeloid-derived suppressor cells to form a premetastatic niche. Serum sEV LOXL2 can reflect a hypoxic and aggressive tumor type and can serve as an alternative to tissue LOXL2 as an independent prognostic factor of overall survival for patients with HNSCC.
Conclusion: sEVs derived from the hypoxic tumor microenvironment of HNSCC can drive local invasion of non-hypoxic HNSCC cells and stimulate premetastatic niche formation by delivering LOXL2 to non-hypoxic HNSCC cells and fibroblasts to induce EMT and fibronectin production, respectively.
Keywords: small extracellular vesicle, premetastatic niche, hypoxia, head and neck squamous cell carcinoma, Lysyl Oxidase Like 2
 Global reach, higher impact
Global reach, higher impact