13.3
Impact Factor
Theranostics 2021; 11(18):9107-9117. doi:10.7150/thno.62444 This issue Cite
Research Paper
Synthesis of precision antibody conjugates using proximity-induced chemistry
1. State Key Laboratory of Chemical Oncogenomics, Key Laboratory of Chemical Genomics, Peking University Shenzhen Graduate School, Shenzhen, 518055, China
2. Department of Chemistry, Rice University, 6100 Main Street, Houston, Texas, 77005, USA
3. Department of Biosciences, Rice University, 6100 Main Street, Houston, Texas, 77005, USA
4. Department of Bioengineering, Rice University, 6100 Main Street, Houston, Texas, 77005, USA
* These authors contributed equally
Abstract
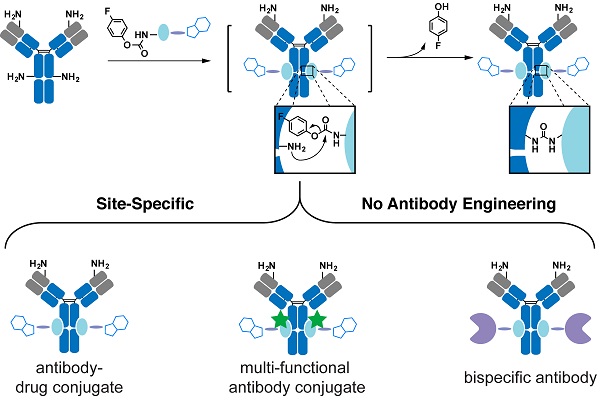
Rationale: Therapeutic antibody conjugates allow for the specific delivery of cytotoxic agents or immune cells to tumors, thus enhancing the antitumor activity of these agents and minimizing adverse systemic effects. Most current antibody conjugates are prepared by nonspecific modification of antibody cysteine or lysine residues, inevitably resulting in the generation of heterogeneous conjugates with limited therapeutic efficacies. Traditional strategies to prepare homogeneous antibody conjugates require antibody engineering or chemical/enzymatic treatments, processes that often affect antibody folding and stability, as well as yield and cost. Developing a simple and cost-effective way to precisely couple functional payloads to native antibodies is of great importance.
Methods: We describe a simple proximity-induced antibody conjugation method (pClick) that enables the synthesis of homogeneous antibody conjugates from native antibodies without requiring additional antibody engineering or post-synthesis treatments. A proximity-activated crosslinker is introduced into a chemically synthesized affinity peptide modified with a bioorthogonal handle. Upon binding to a specific antibody site, the affinity peptide covalently attaches to the antibody via spontaneous crosslinking, yielding an antibody molecule ready for bioorthogonal conjugation with payloads.
Results: We have prepared well-defined antibody-drug conjugates and bispecific small molecule-antibody conjugates using pClick technology. The resulting conjugates exhibit excellent in vitro cytotoxic activity against cancer cells and, in the case of bispecific conjugates, superb antitumor activity in mouse xenograft models.
Conclusions: Our pClick technology enables efficient, simple, and site-specific conjugation of various moieties to the existing native antibodies. This technology does not require antibody engineering or additional UV/chemical/enzymatic treatments, therefore providing a general, convenient strategy for developing novel antibody conjugates.
Keywords: Antibody-drug conjugates, Bispecific antibodies, Proximity-Induced Chemistry, Site-specific conjugation, Antibody Conjugation
 Global reach, higher impact
Global reach, higher impact