Impact Factor
Theranostics 2021; 11(18):9089-9106. doi:10.7150/thno.62724 This issue Cite
Research Paper
Monoethanolamine-induced glucose deprivation promotes apoptosis through metabolic rewiring in prostate cancer
1. Department of Biology, Georgia State University, Atlanta, GA, USA-30303
2. Caris Life Sciences, Tempe, Arizona, USA-85282
3. Department of Chemistry, Indian Institute of Technology Bombay, Powai, Mumbai, India-400076
4. Department of Pathology & Laboratory Medicine, Emory University School of Medicine, Atlanta, GA, USA-30324
#Authors contributed equally
Abstract
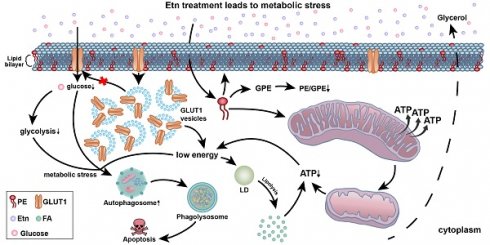
Rationale: Cancer cells rely on glucose metabolism for fulfilling their high energy demands. We previously reported that monoethanolamine (Etn), an orally deliverable lipid formulation, reduced intracellular glucose and glutamine levels in prostate cancer (PCa). Glucose deprivation upon Etn treatment exacerbated metabolic stress in PCa, thereby enhancing cell death. Moreover, Etn was potent in inhibiting tumor growth in a PCa xenograft model. However, the precise mechanisms underlying Etn-induced metabolic stress in PCa remain elusive. The purpose of the present study was to elucidate the mechanisms contributing to Etn-mediated metabolic rewiring in PCa.
Methods: Glucose transporters (GLUTs) facilitate glucose transport across the plasma membrane. Thus, we assessed the expression of GLUTs and the internalization of GLUT1 in PCa. We also evaluated the effects of Etn on membrane dynamics, mitochondrial structure and function, lipid droplet density, autophagy, and apoptosis in PCa cells.
Results: Compared to other GLUTs, GLUT1 was highly upregulated in PCa. We observed enhanced GLUT1 internalization, altered membrane dynamics, and perturbed mitochondrial structure and function upon Etn treatment. Etn-induced bioenergetic stress enhanced lipolysis, decreased lipid droplet density, promoted accumulation of autophagosomes, and increased apoptosis.
Conclusion: We provide the first evidence that Etn alters GLUT1 trafficking leading to metabolic stress in PCa. By upregulating phosphatidylethanolamine (PE), Etn modulates membrane fluidity and affects mitochondrial structure and function. Etn also induces autophagy in PCa cells, thereby promoting apoptosis. These data strongly suggest that Etn rewires cellular bioenergetics and could serve as a promising anticancer agent for PCa.
Keywords: prostate cancer, monoethanolamine, metabolism, autophagy, apoptosis
 Global reach, higher impact
Global reach, higher impact