Impact Factor
Theranostics 2021; 11(18):8909-8925. doi:10.7150/thno.61651 This issue Cite
Research Paper
A tumor microenvironment responsive nanosystem for chemodynamic/chemical synergistic theranostics of colorectal cancer
1. Department of Radiology, Sir Run Run Shaw Hospital, Zhejiang University School of Medicine, Hangzhou, 310016, China.
2. Department of General Surgery, Sir Run Run Shaw Hospital, Zhejiang University School of Medicine, Hangzhou, 310016, China.
3. MOE Key Laboratory of Macromolecular Synthesis and Functionalization, Department of Polymer Science and Engineering, Zhejiang University, Hangzhou, 310027, China.
4. Cancer Center, Zhejiang University, Hangzhou, Zhejiang, 310058, China.
Abstract
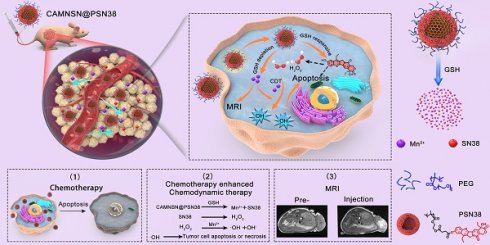
Rationale: The synergism of new modalities alongside chemodynamic therapy into common chemotherapy has shown promising potential in clinical applications. This paper reports a tumor microenvironment-responsive nanosystem for chemodynamic/chemical synergistic therapy and magnetic resonance imaging (MRI).
Methods: The biodegradable nanosystem is synthesized using a surface-modified chain transfer agent for surface-initiated living radical polymerization of the chemotherapeutic drug.
Results: In this nanosystem, named CAMNSN@PSN38, the cycling time and solubility of the chemotherapeutic drug are improved. The nanoparticles delivered to tumor tissues gradually release the chemotherapeutic drug and Mn2+ through glutathione (GSH)-triggered biodegradation in the tumor microenvironment. SN38, the released chemotherapeutic drug, not only shows excellent chemical therapy effects but also improves the generation of H2O2. Furthermore, with the Fenton-like agent Mn2+, the generation of reactive oxygen species (ROS) is improved markedly. Finally, CAMNSN@PSN38 shows excellent inhibition of tumor growth in three colorectal cancer tumor models, with an improved accumulation of ROS and controlled release of SN38.
Conclusions: The CAMNSN@PSN38-mediated chemodynamic/chemical synergistic therapy provides a promising paradigm for the treatment and MRI-guided therapy of colorectal cancer.
Keywords: Chemodynamic therapy, Nanotheranostics, Microenvironment-responsive, Magnetic resonance imaging, Synergistic therapy
 Global reach, higher impact
Global reach, higher impact