Impact Factor
Theranostics 2021; 11(18):8874-8893. doi:10.7150/thno.59759 This issue Cite
Research Paper
Macrophage targeted theranostic strategy for accurate detection and rapid stabilization of the inflamed high-risk plaque
1. Multimodal Imaging and Theranostic Lab., Cardiovascular Center, Korea University Guro Hospital, Seoul, South Korea.
2. Department of Mechanical Engineering, Korea Advanced Institute of Science and Technology, Daejeon, South Korea.
3. Department of Systems Biotechnology, Chung-Ang University, Anseong, South Korea.
#These authors contributed equally to this work.
Abstract
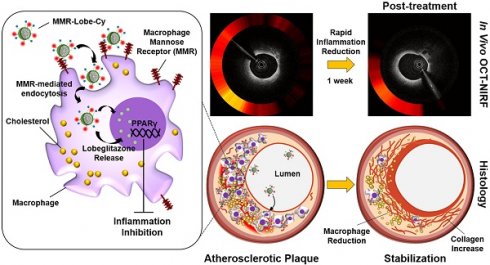
Rationale: Inflammation plays a pivotal role in the pathogenesis of the acute coronary syndrome. Detecting plaques with high inflammatory activity and specifically treating those lesions can be crucial to prevent life-threatening cardiovascular events.
Methods: Here, we developed a macrophage mannose receptor (MMR)-targeted theranostic nanodrug (mannose-polyethylene glycol-glycol chitosan-deoxycholic acid-cyanine 7-lobeglitazone; MMR-Lobe-Cy) designed to identify inflammatory activity as well as to deliver peroxisome proliferator-activated gamma (PPARγ) agonist, lobeglitazone, specifically to high-risk plaques based on the high mannose receptor specificity. The MMR-Lobe-Cy was intravenously injected into balloon-injured atheromatous rabbits and serial in vivo optical coherence tomography (OCT)-near-infrared fluorescence (NIRF) structural-molecular imaging was performed.
Results: One week after MMR-Lobe-Cy administration, the inflammatory NIRF signals in the plaques notably decreased compared to the baseline whereas the signals in saline controls even increased over time. In accordance with in vivo imaging findings, ex vivo NIRF signals on fluorescence reflectance imaging (FRI) and plaque inflammation by immunostainings significantly decreased compared to oral lobeglitazone group or saline controls. The anti-inflammatory effect of MMR-Lobe-Cy was mediated by inhibition of TLR4/NF-κB pathway. Furthermore, acute resolution of inflammation altered the inflamed plaque into a stable phenotype with less macrophages and collagen-rich matrix.
Conclusion: Macrophage targeted PPARγ activator labeled with NIRF rapidly stabilized the inflamed plaques in coronary sized artery, which could be quantitatively assessed using intravascular OCT-NIRF imaging. This novel theranostic approach provides a promising theranostic strategy for high-risk coronary plaques.
Keywords: atherosclerosis, targeted theranostics, drug delivery, PPARγ, OCT-NIRF
 Global reach, higher impact
Global reach, higher impact