Impact Factor
Theranostics 2021; 11(18):8797-8812. doi:10.7150/thno.61601 This issue Cite
Research Paper
Activating AhR alleviates cognitive deficits of Alzheimer's disease model mice by upregulating endogenous Aβ catabolic enzyme Neprilysin
1. Sir Run run Hospital, Nanjing Medical University, Nanjing, China.
2. State Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing, China.
Abstract
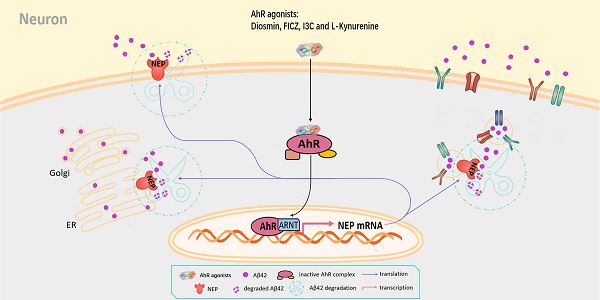
Rationale: Neprilysin (NEP) is a major endogenous catabolic enzyme of amyloid β (Aβ). Previous studies have suggested that increasing NEP expression in animal models of Alzheimer's disease had an ameliorative effect. However, the underlying signaling pathway that regulates NEP expression remains unclear. The aryl hydrocarbon receptor (AhR) is a ligand-activated cytoplasmic receptor and transcription factor. Recent studies have shown that AhR plays essential roles in the central nervous system (CNS), but its physiological and pathological roles in regulating NEP are not entirely known.
Methods: Western blotting, immunofluorescence, quantitative RT-PCR and enzyme activity assay were used to verify the effects of AhR agonists on NEP in a cell model (N2a) and a mouse model (APP/PS1). Luciferase reporter assay and chromatin immunoprecipitation (ChIP) assay were conducted to investigate the roles of AhR in regulating NEP transcription. Object recognition test and the Morris water maze task were performed to assess the cognitive capacity of the mice.
Results: Activating AhR by the endogenous ligand L-Kynurenine (L-KN) or FICZ, or by the exogenous ligand diosmin or indole-3-carbinol (I3C) significantly increases NEP expression and enzyme activity in N2a cells and APP/PS1 mice. We also found that AhR is a direct transcription factor of NEP. Diosmin treatment effectively ameliorated the cognitive disorder and memory deficit of APP/PS1 transgenic mice. By knocking down AhR or using a small molecular inhibitor targeting AhR or NEP, we found that diosmin enhanced Aβ degradation through activated AhR and increased NEP expression.
Conclusions: These results indicate a novel pathway for regulating NEP expression in neurons and that AhR may be a potential therapeutic target for the treatment of Alzheimer's disease.
Keywords: Neprilysin, Aryl hydrocarbon receptor, Anti-amyloidogenic therapy, Amyloid-β, Alzheimer's disease
 Global reach, higher impact
Global reach, higher impact