Impact Factor
Theranostics 2021; 11(18):8771-8796. doi:10.7150/thno.62642 This issue Cite
Review
Effective tools for RNA-derived therapeutics: siRNA interference or miRNA mimicry
1. Cardiovascular Research Institute, Yong Loo Lin School of Medicine, National University of Singapore, 117599 Singapore.
2. Department of Medicine, National University Health System, 119228 Singapore.
3. Christchurch Heart Institute, Department of Medicine, University of Otago Christchurch, New Zealand.
Abstract
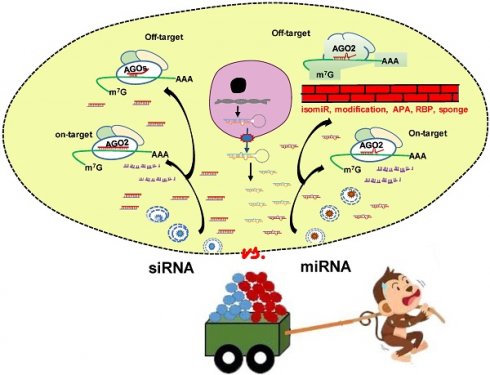
The approval of the first small interfering RNA (siRNA) drug Patisiran by FDA in 2018 marks a new era of RNA interference (RNAi) therapeutics. MicroRNAs (miRNA), an important post-transcriptional gene regulator, are also the subject of both basic research and clinical trials. Both siRNA and miRNA mimics are ~21 nucleotides RNA duplexes inducing mRNA silencing. Given the well performance of siRNA, researchers ask whether miRNA mimics are unnecessary or developed siRNA technology can pave the way for the emergence of miRNA mimic drugs. Through comprehensive comparison of siRNA and miRNA, we focus on (1) the common features and lessons learnt from the success of siRNAs; (2) the unique characteristics of miRNA that potentially offer additional therapeutic advantages and opportunities; (3) key areas of ongoing research that will contribute to clinical application of miRNA mimics. In conclusion, miRNA mimics have unique properties and advantages which cannot be fully matched by siRNA in clinical applications. MiRNAs are endogenous molecules and the gene silencing effects of miRNA mimics can be regulated or buffered to ameliorate or eliminate off-target effects. An in-depth understanding of the differences between siRNA and miRNA mimics will facilitate the development of miRNA mimic drugs.
Keywords: RNA interference (RNAi), therapeutics, siRNA, miRNA, off-target effect
 Global reach, higher impact
Global reach, higher impact