13.3
Impact Factor
Theranostics 2021; 11(18):8660-8673. doi:10.7150/thno.62820 This issue Cite
Research Paper
Exosomal miR-21 from tubular cells contributes to renal fibrosis by activating fibroblasts via targeting PTEN in obstructed kidneys
1. Department of Urology, Renmin Hospital of Wuhan University, Wuhan, 430060, China
2. Department of Anesthesiology, Renmin Hospital of Wuhan University, Wuhan, 430060, China
*Sheng Zhao and Wei Li contributed equally to this work.
#Fan Cheng and Xiangjun Zhou contributed equally to this work.
Abstract
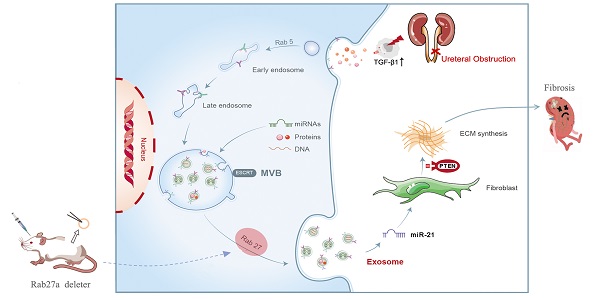
Rationale: Ureteral obstruction-induced hydronephrosis is associated with renal fibrosis and progressive chronic kidney disease (CKD). Exosome-mediated cell-cell communication has been suggested to be involved in various diseases, including renal fibrosis. However, little is known regarding how exosomes regulate renal fibrosis in obstructed kidneys.
Methods: We first examined the secretion of exosomes in UUO (unilateral ureteral obstruction) mouse kidneys and TGF-β1-stimulated tubular epithelial cells (NRK-52E). Exosomes from NRK-52E cells were subsequently harvested and incubated with fibroblasts (NRK-49F) or injected into UUO mice via the tail vein. We next constructed Rab27a knockout mice to further confirm the role of exosome-mediated epithelial-fibroblast communication relevant to renal fibrosis in UUO mice. High-throughput miRNA sequencing was performed to detect the miRNA profiles of TGFβ1-Exos. The roles of candidate miRNAs, their target genes and relevant pathways were predicted and assessed in vitro and in vivo by setting specific miRNA mimic, miRNA inhibitor, siRNA or miRNA LNA groups.
Results: Increased renal fibrosis was associated with prolonged UUO days, and the secretion of exosomes was markedly increased in UUO kidneys and TGF-β1-stimulated NRK-52E cells. Purified exosomes from TGF-β1-stimulated NRK-52E cells could activate fibroblasts and aggravate renal fibrosis in vitro and in vivo. In addition, the inhibition of exosome secretion by Rab27a knockout or GW4869 treatment abolished fibroblast activation and ameliorated renal fibrosis. Exosomal miR-21 was significantly increased in TGFβ1-Exos compared with Ctrl-Exos, and PTEN is a certain target of miR-21. The promotion or inhibition of epithelial exosomal miR-21 correspondingly accelerated or abolished fibroblast activation in vitro, and renal fibrosis after UUO was alleviated by miR-21-deficient exosomes in vivo through the PTEN/Akt pathway.
Conclusion: Our findings reveal that exosomal miR-21 from tubular epithelial cells may accelerate the development of renal fibrosis by activating fibroblasts via the miR-21/PTEN/Akt pathway in obstructed kidneys.
Keywords: renal fibrosis, unilateral ureteral obstruction, exosome, microRNA
 Global reach, higher impact
Global reach, higher impact