13.3
Impact Factor
Theranostics 2021; 11(18):8640-8659. doi:10.7150/thno.61833 This issue Cite
Research Paper
Inhibiting microglia proliferation after spinal cord injury improves recovery in mice and nonhuman primates
1. MMDN, Univ. Montpellier, EPHE, INSERM, Montpellier, France; Department of Neurosurgery, CHU, Montpellier, France.
2. MMDN, Univ. Montpellier, EPHE, INSERM, Montpellier, France.
3. MMDN, Univ Montpellier, EPHE, INSERM, Montpellier, France; PSL Research University, Paris, France.
4. University of Montpellier, UMR 5221 CNRS, Montpellier, France.
5. INSERM U1051, Institute for Neurosciences of Montpellier, Montpellier, France.
6. Montpellier Resources Imaging (MRI), Montpellier, France.
7. Institut Universitaire de France (IUF).
#These authors contributed equally to this work.
Abstract
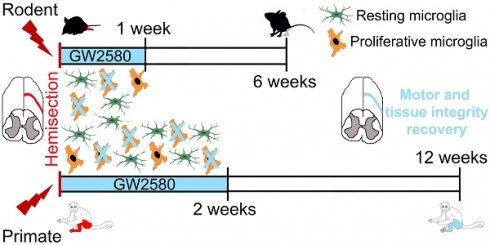
No curative treatment is available for any deficits induced by spinal cord injury (SCI). Following injury, microglia undergo highly diverse activation processes, including proliferation, and play a critical role on functional recovery.
In a translational objective, we investigated whether a transient pharmacological reduction of microglia proliferation after injury is beneficial for functional recovery after SCI in mice and nonhuman primates.
Methods: The colony stimulating factor-1 receptor (CSF1R) regulates proliferation, differentiation, and survival of microglia. We orally administrated GW2580, a CSF1R inhibitor that inhibits microglia proliferation. In mice and nonhuman primates, we then analyzed treatment outcomes on locomotor function and spinal cord pathology. Finally, we used cell-specific transcriptomic analysis to uncover GW2580-induced molecular changes in microglia.
Results: First, transient post-injury GW2580 administration in mice improves motor function recovery, promotes tissue preservation and/or reorganization (identified by coherent anti-stokes Raman scattering microscopy), and modulates glial reactivity.
Second, post-injury GW2580-treatment in nonhuman primates reduces microglia proliferation, improves motor function recovery, and promotes tissue protection.
Finally, GW2580-treatment in mice induced down-regulation of proliferation-associated transcripts and inflammatory associated genes in microglia that may account for reduced neuroinflammation and improved functional recovery following SCI.
Conclusion: Thus, a transient oral GW2580 treatment post-injury may provide a promising therapeutic strategy for SCI patients and may also be extended to other central nervous system disorders displaying microglia activation.
Keywords: spinal cord injury, microglia, proliferation, rodent, primates
 Global reach, higher impact
Global reach, higher impact