13.3
Impact Factor
Theranostics 2021; 11(16):7755-7766. doi:10.7150/thno.51419 This issue Cite
Research Paper
Molecular imaging of fibroblast activation protein after myocardial infarction using the novel radiotracer [68Ga]MHLL1
1. Department of Nuclear Medicine, Hannover Medical School
2. Department of Cardiology and Angiology, Hannover Medical School
3. Molecular Imaging and Radiochemistry, Department of Clinical Radiology and Nuclear Medicine, Medical Faculty Mannheim of Heidelberg University
§ equally contributing first authors;
$ equally contributing senior authors
Abstract
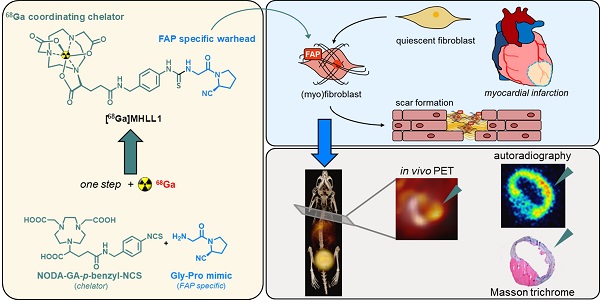
Background: Myocardial infarction (MI) evokes an organized remodeling process characterized by the activation and transdifferentiation of quiescent cardiac fibroblasts to generate a stable collagen rich scar. Early fibroblast activation may be amenable to targeted therapy, but is challenging to identify in vivo. We aimed to non-invasively image active fibrosis by targeting the fibroblast activation protein (FAP) expressed by activated (myo)fibroblasts, using a novel positron emission tomography (PET) radioligand [68Ga]MHLL1 after acute MI.
Methods: One-step chemical synthesis and manual as well as module-based radiolabeling yielded [68Ga]MHLL1. Binding characteristics were evaluated in murine and human FAP-transfected cells, and stability tested in human serum. Biodistribution in healthy animals was interrogated by dynamic PET imaging, and metabolites were measured in blood and urine. The temporal pattern of FAP expression was determined by serial PET imaging at 7 d and 21 d after coronary artery ligation in mice as percent injected dose per gram (%ID/g). PET measurements were validated by ex vivo autoradiography and immunostaining for FAP and inflammatory macrophages.
Results: [68Ga]MHLL1 displayed specific uptake in murine and human FAP-positive cells (p = 0.0208). In healthy mice the tracer exhibited favorable imaging characteristics, with low blood pool retention and dominantly renal clearance. At 7 d after coronary artery ligation, [68Ga]MHLL1 uptake was elevated in the infarct relative to the non-infarcted remote myocardium (1.3 ± 0.3 vs. 1.0 ± 0.2 %ID/g, p < 0.001) which persisted to 21 d after MI (1.3 ± 0.4 vs. 1.1 ± 0.4 %ID/g, p = 0.013). Excess unlabeled compound blocked tracer accumulation in both infarct and non-infarct remote myocardium regions (p < 0.001). Autoradiography and histology confirmed the regional uptake of [68Ga]MHLL1 in the infarct and especially border zone regions, as identified by Masson trichrome collagen staining. Immunostaining further delineated persistent FAP expression at 7 d and 21 d post-MI in the border zone, consistent with tracer distribution in vivo.
Conclusion: The simplified synthesis of [68Ga]MHLL1 bears promise for non-invasive characterization of fibroblast activation protein early in remodeling after MI.
 Global reach, higher impact
Global reach, higher impact