Impact Factor
Theranostics 2021; 11(16):7620-7639. doi:10.7150/thno.58515 This issue Cite
Research Paper
Fiber scaffold bioartificial liver therapy relieves acute liver failure and extrahepatic organ injury in pigs
1. Department of Hepatobiliary Surgery II, Guangdong Provincial Research Center for Artificial Organ and Tissue Engineering, Guangzhou Clinical Research and Transformation Center for Artificial Liver, Institute of Regenerative Medicine, Zhujiang Hospital of Southern Medical University, Guangzhou 510515, China.
2. State Key Laboratory of Organ Failure Research, Southern Medical University, Guangzhou 510515, China.
3. Department of Medical Oncology, Jinling Hospital, First School of Clinical Medicine, Southern Medical University, Nanjing 210000, China.
4. Guangdong Qianhui Biotechnology Co., Ltd., Guangzhou 510285, China.
*These authors contributed equally to this work.
Abstract
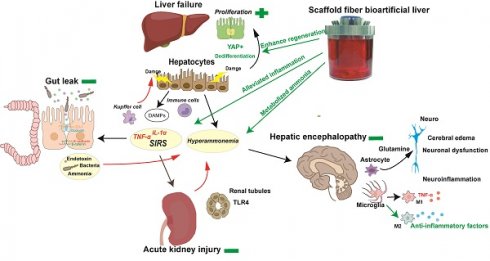
Rationale: Acute liver failure (ALF) causes severe liver injury and a systemic inflammatory response, leading to multiorgan failure with a high short-term mortality. Bioartificial liver (BAL) therapy is a promising approach that is hampered by the lack of appropriate bioreactors and carriers to retain hepatic cell function and poor understanding of BAL treatment mechanisms in ALF and extrahepatic organ injury. Recently, we used a fiber scaffold bioreactor (FSB) for the high-density, three-dimensional (3D) culture of primary porcine hepatocytes (PPHs) combined with an absorption component to construct a BAL and verified its function in a D-galactosamine (D-gal)-induced ALF porcine model to evaluate its protective effects on the liver and extrahepatic organs.
Methods: Male pigs were randomized into standard/supportive therapy (ST), ST+no-cell BAL (ST+Sham BAL) and ST+BAL groups and received treatment 48 h after receiving a D-gal injection. Changes in blood chemistry and clinical symptoms were monitored for 120 h. Tissues and plasma were collected for analysis by pathological examination, immunoblotting, quantitative PCR and immunoassays.
Results: PPHs cultured in the FSB obtained sufficient aeration and nutrition for high-density, 3D culture and maintained superior viability and functionality (biosynthesis and detoxification) compared with those cultured in flasks. All the animals developed ALF, acute kidney injury (AKI) and hepatic encephalopathy (HE) 48 h after D-gal infusion and received corresponding therapies. Animals in the BAL group showed markedly improved survival (4/5; 80%) compared with those in the ST+Sham BAL (0/5; p < 0.001) and ST (0/5; p < 0.001) groups. The levels of blood ammonia and biochemical and inflammatory indices were alleviated after BAL treatment. Increased liver regeneration and attenuations in the occurrence and severity of ALF, AKI and HE were observed in the ST+BAL group compared with the ST (p = 0.0009; p = 0.038) and ST+Sham BAL (p = 0.011; p = 0.031) groups. Gut leakage, the plasma endotoxin level, bacterial translocation, and peripheral and neuroinflammation were alleviated in the ST+BAL group compared with those in the other groups.
Conclusions: BAL treatment enhanced liver regeneration and alleviated the systemic inflammatory response and extrahepatic organ injury to prolong survival in the ALF model and has potential as a therapeutic approach for ALF patients.
Keywords: liver failure, bioartificial liver, regeneration, multiorgan failure, hepatic encephalopathy
 Global reach, higher impact
Global reach, higher impact