13.3
Impact Factor
Theranostics 2021; 11(14):6860-6872. doi:10.7150/thno.57511 This issue Cite
Research Paper
Magnetism-mediated targeting hyperthermia-immunotherapy in “cold” tumor with CSF1R inhibitor
1. Artemisinin Research Center, Guangzhou University of Chinese Medicine, 12 Jichang Rd, Guangzhou 510450, China.
2. State Key Laboratory of Drug Research, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, 501 Haike Rd, Shanghai 201203, China.
3. University of Chinese Academy of Sciences, Beijing, China.
4. Zhongshan Institute for Drug Discovery, The Institutes of Drug Discovery and Development, CAS, Zhongshan 528437, China.
5. NMPA Key Laboratory for Quality Research and Evaluation of Pharmaceutical Excipients, Shanghai 201203, China.
#Equal contributions to this work.
Abstract
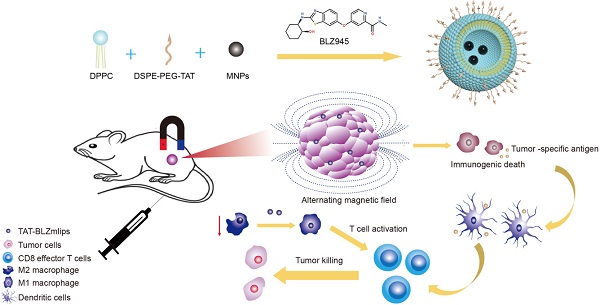
Background: Immunotherapy has profoundly changed the landscape of cancer management and represented the most significant breakthrough. Yet, it is a formidable challenge that the majority of cancers - the so-called “cold” tumors - poorly respond to immunotherapy. To find a general immunoregulatory modality that can be applied to a broad spectrum of cancers is an urgent need.
Methods: Magnetic hyperthermia (MHT) possesses promise in cancer therapy. We develop a safe and effective therapeutic strategy by using magnetism-mediated targeting MHT-immunotherapy in “cold” colon cancer. A magnetic liposomal system modified with cell-penetrating TAT peptide was developed for targeted delivery of a CSF1R inhibitor (BLZ945), which can block the CSF1-CSF1R pathway and reduce M2 macrophages. The targeted delivery strategy is characterized by its magnetic navigation and TAT-promoting intratumoral penetration.
Results: The liposomes (termed TAT-BLZmlips) can induce ICD and cause excessive CRT exposure on the cell surface, which transmits an “eat-me” signal to DCs to elicit immunity. The combination of MHT and BLZ945 can repolarize M2 macrophages in the tumor microenvironment to relieve immunosuppression, normalize the tumor blood vessels, and promote T-lymphocyte infiltration. The antitumor effector CD8+ T cells were increased after treatment.
Conclusion: This work demonstrated that TAT-BLZmlips with magnetic navigation and MHT can remodel tumor microenvironment and activate immune responses and memory, thus inhibiting tumor growth and recurrence.
Keywords: magnetic hyperthermia, CSF1R inhibitor, tumor microenvironment, tumor-associated macrophage, immune memory, targeted delivery, magnetic liposomes
 Global reach, higher impact
Global reach, higher impact