13.3
Impact Factor
Theranostics 2021; 11(14):6800-6817. doi:10.7150/thno.56989 This issue Cite
Research Paper
Radionuclide-based molecular imaging allows CAR-T cellular visualization and therapeutic monitoring
1. Department of Nuclear Medicine, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, China.
2. Hubei Key Laboratory of Molecular Imaging, Wuhan 430022, China.
3. Department of nuclear medicine, Zigong First People's Hospital, Zigong 643000, China.
4. Nuclear Medicine and Molecular Imaging Key Laboratory of Sichuan Province, Luzhou 646000, China.
5. Department of Immunology, School of Basic Medicine, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, China.
Abstract
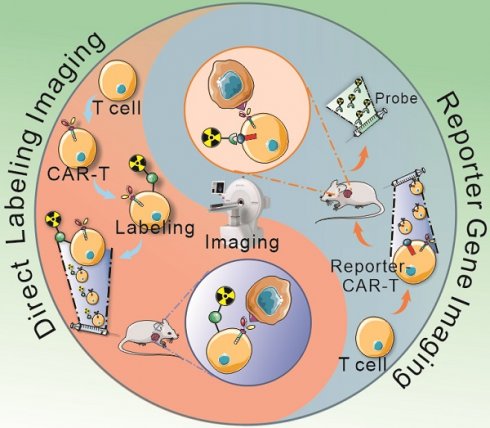
Chimeric antigen receptor T cell (CAR-T) therapy is a new and effective form of adoptive cell therapy that is rapidly entering the mainstream for the treatment of CD19-positive hematological cancers because of its impressive effect and durable responses. Huge challenges remain in achieving similar success in patients with solid tumors. The current methods of monitoring CAR-T, including morphological imaging (CT and MRI), blood tests, and biopsy, have limitations to assess whether CAR-T cells are homing to tumor sites and infiltrating into tumor bed, or to assess the survival, proliferation, and persistence of CAR-T cells in solid tumors associated with an immunosuppressive microenvironment. Radionuclide-based molecular imaging affords improved CAR-T cellular visualization and therapeutic monitoring through either a direct cellular radiolabeling approach or a reporter gene imaging strategy, and endogenous cell imaging is beneficial to reflect functional information and immune status of T cells. Focusing on the dynamic monitoring and precise assessment of CAR-T therapy, this review summarizes the current applications of radionuclide-based noninvasive imaging in CAR-T cells visualization and monitoring and presents current challenges and strategic choices.
Keywords: chimeric antigen receptor T cell, molecular imaging, side effects, therapeutic monitoring, direct labeling, reporter gene, endogenous cell
 Global reach, higher impact
Global reach, higher impact