13.3
Impact Factor
Theranostics 2021; 11(14):6668-6681. doi:10.7150/thno.56494 This issue Cite
Research Paper
Intratumoral expression of interleukin 23 variants using oncolytic vaccinia virus elicit potent antitumor effects on multiple tumor models via tumor microenvironment modulation
1. Allegheny Health Network Cancer Institute, Pittsburgh, PA 15212, USA.
2. Department of Surgery, Drexel University College of Medicine, Philadelphia, PA 19104, USA.
3. UPMC Hillman Cancer Center and Department of Surgery, University of Pittsburgh School of Medicine, Pittsburgh, PA 15213, USA.
4. Cancer Center, Union Hospital, Huazhong University of Science and Technology, Wuhan, Hubei Province, China.
5. Shanghai Jiao Tong University Affiliated Sixth People's Hospital, Shanghai, China.
6. Department of Cancer Center, Renmin Hospital of Wuhan University, Wuhan, Hubei Province, China.
7. Department of Immunology, School of Biology and Basic Medical Sciences, Medical College, Soochow University, Suzhou, Jiangsu Province, China.
8. Department of Oncology and Hematology, China-Japan Union Hospital of Jilin University, Changchun, Jilin Province, China.
9. Xiangya School of Medicine, Central South University, Changsha, Hunan Province, China.
* These authors contributed equally to this work.
Abstract
Background: Newly emerging cancer immunotherapy has led to significant progress in cancer treatment; however, its efficacy is limited in solid tumors since the majority of them are “cold” tumors. Oncolytic viruses, especially when properly armed, can directly target tumor cells and indirectly modulate the tumor microenvironment (TME), resulting in “hot” tumors. These viruses can be applied as a cancer immunotherapy approach either alone or in combination with other cancer immunotherapies. Cytokines are good candidates to arm oncolytic viruses. IL-23, an IL-12 cytokine family member, plays many roles in cancer immunity. Here, we used oncolytic vaccinia viruses to deliver IL-23 variants into the tumor bed and explored their activity in cancer treatment on multiple tumor models.
Methods: Oncolytic vaccinia viruses expressing IL-23 variants were generated by homologue recombination. The characteristics of these viruses were in vitro evaluated by RT-qPCR, ELISA, flow cytometry and cytotoxicity assay. The antitumor effects of these viruses were evaluated on multiple tumor models in vivo and the mechanisms were investigated by RT-qPCR and flow cytometry.
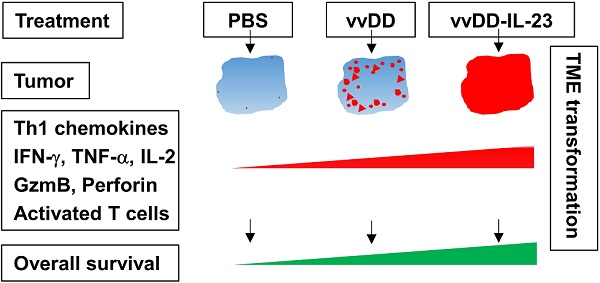
Results: IL-23 prolonged viral persistence, probably mediated by up-regulated IL-10. The sustainable IL-23 expression and viral oncolysis elevated the expression of Th1 chemokines and antitumor factors such as IFN-γ, TNF-α, Perforin, IL-2, Granzyme B and activated T cells in the TME, transforming the TME to be more conducive to antitumor immunity. This leads to a systemic antitumor effect which is dependent on CD8+ and CD4+ T cells and IFN-γ. Oncolytic vaccinia viruses could not deliver stable IL-23A to the tumor, attributed to the elevated tristetraprolin which can destabilize the IL-23A mRNA after the viral treatment; whereas vaccinia viruses could deliver membrane-bound IL-23 to elicit a potent antitumor effect which might avoid the possible toxicity normally associated with systemic cytokine exposure.
Conclusion: Either secreted or membrane-bound IL-23-armed vaccinia virus can induce potent antitumor effects and IL-23 is a candidate cytokine to arm oncolytic viruses for cancer immunotherapy.
Keywords: oncolytic virus, IL-23, cancer immunotherapy, tumor microenvironment, efficacy.
 Global reach, higher impact
Global reach, higher impact