13.3
Impact Factor
Theranostics 2021; 11(12):6044-6057. doi:10.7150/thno.55092 This issue Cite
Research Paper
Repurposing 18F-FMISO as a PET tracer for translational imaging of nitroreductase-based gene directed enzyme prodrug therapy
1. Centre for Cancer Biomarkers CCBIO, Department of Clinical Science, University of Bergen, Jonas Lies vei 65, N-5021, Bergen, Norway.
2. University Hospital 12 de Octubre, Madrid, Spain; Lung Cancer Unit H12O-CNIO, Spain.
3. Department of Chemistry and Centre for Pharmacy, University of Bergen, Allégaten 41, N-5007, Bergen, Norway.
4. KinN Therapeutics AS, Jonas Lies vei 65, 5021 Bergen, Norway.
5. Department of Pathology, Oslo University Hospital, Norwegian Radium Hospital, and Faculty of Medicine, Institute of Clinical Medicine, University of Oslo, Oslo, Norway.
6. Molecular imaging Center, Department of Biomedicine, University of Bergen, Jonas Lies vei 91, N-5009, Bergen, Norway.
7. Western Norway University College, Inndalsveien 28, N-5063, Bergen, Norway.
8. Department of Pathology, Haukeland University Hospital, Jonas Lies vei 65, N-5021, Bergen, Norway.
9. Centre for Pharmacy, Department of Clinical Science, University of Bergen, Jonas Lies vei 65, Bergen 5021, Norway.
10. Vivarium, Department of Clinical Medicine, University of Bergen, Jonas Lies vei 65, 5021 Bergen, Norway.
*These authors contributed equally to this work.
Abstract
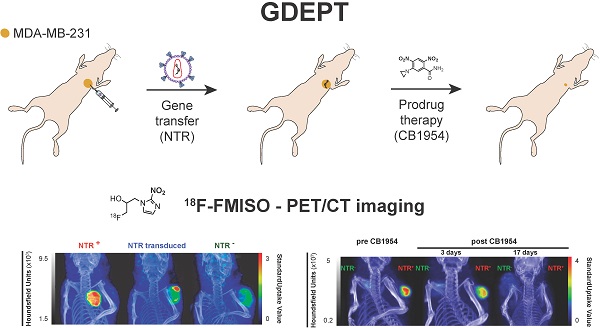
Nitroreductases (NTR) are a family of bacterial enzymes used in gene directed enzyme prodrug therapy (GDEPT) that selectively activate prodrugs containing aromatic nitro groups to exert cytotoxic effects following gene transduction in tumours. The clinical development of NTR-based GDEPT has, in part, been hampered by the lack of translational imaging modalities to assess gene transduction and drug cytotoxicity, non-invasively. This study presents translational preclinical PET imaging to validate and report NTR activity using the clinically approved radiotracer, 18F-FMISO, as substrate for the NTR enzyme.
Methods: The efficacy with which 18F-FMISO could be used to report NfsB NTR activity in vivo was investigated using the MDA-MB-231 mammary carcinoma xenograft model. For validation, subcutaneous xenografts of cells constitutively expressing NTR were imaged using 18F-FMISO PET/CT and fluorescence imaging with CytoCy5S, a validated fluorescent NTR substrate. Further, examination of the non-invasive functionality of 18F-FMISO PET/CT in reporting NfsB NTR activity in vivo was assessed in metastatic orthotopic NfsB NTR expressing xenografts and metastasis confirmed by bioluminescence imaging. 18F-FMISO biodistribution was acquired ex vivo by an automatic gamma counter measuring radiotracer retention to confirm in vivo results. To assess the functional imaging of NTR-based GDEPT with 18F-FMISO, PET/CT was performed to assess both gene transduction and cytotoxicity effects of prodrug therapy (CB1954) in subcutaneous models.
Results: 18F-FMISO retention was detected in NTR+ subcutaneous xenografts, displaying significantly higher PET contrast than NTR- xenografts (p < 0.0001). Substantial 18F-FMISO retention was evident in metastases of orthotopic xenografts (p < 0.05). Accordingly, higher 18F-FMISO biodistribution was prevalent ex vivo in NTR+ xenografts. 18F-FMISO NfsB NTR PET/CT imaging proved useful for monitoring in vivo NTR transduction and the cytotoxic effect of prodrug therapy.
Conclusions: 18F-FMISO NfsB NTR PET/CT imaging offered significant contrast between NTR+ and NTR- tumours and effective resolution of metastatic progression. Furthermore, 18F-FMISO NfsB NTR PET/CT imaging proved efficient in monitoring the two steps of GDEPT, in vivo NfsB NTR transduction and response to CB1954 prodrug therapy. These results support the repurposing of 18F-FMISO as a readily implementable PET imaging probe to be employed as companion diagnostic test for NTR-based GDEPT systems.
Keywords: 18F-FMISO, Gene-directed enzymatic prodrug therapy, GDEPT, nitroreductase, NTR, cancer, xenograft, preclinical, mouse, PET/CT, imaging
 Global reach, higher impact
Global reach, higher impact