13.3
Impact Factor
Theranostics 2021; 11(5):2410-2423. doi:10.7150/thno.47182 This issue Cite
Research Paper
Platelet-derived microvesicles induce calcium oscillations and promote VSMC migration via TRPV4
1. Institute of Mechanobiology& Medical Engineering, School of Life Sciences &Biotechnology, Shanghai Jiao Tong University, Shanghai, China
2. School of Perfume and Aroma Technology, Shanghai Institute of Technology, Shanghai, China
3. Department of Bioengineering, Institute of Engineering in Medicine, University of California, San Diego, San Diego, United States
4. Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, School of Biological Science and Medical Engineering, Beihang University, Beijing, 100083, China
5. Beijing Advanced Innovation Center for Biomedical Engineering, Beihang University, Beijing, 100083, China
# These authors contributed equally to this manuscript.
Abstract
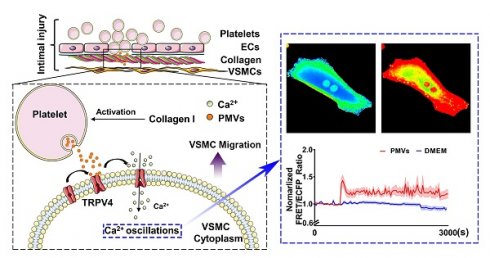
Rationale: Abnormal migration of vascular smooth muscle cells (VSMCs) from the media to the interior is a critical process during the intimal restenosis caused by vascular injury. Here, we determined the role of platelet-derived microvesicles (PMVs) released by activated platelets in VSMC migration.
Methods: A percutaneous transluminal angioplasty balloon dilatation catheter was used to establish vascular intimal injury. Collagen I was used to activate PMVs, mimicking collagen exposure during intimal injury. To determine the effects of PMVs on VSMC migration in vitro, scratch wound healing assays were performed. Fluorescence resonance energy transfer was used to detect variations of calcium dynamics in VSMCs.
Results: Morphological results showed that neointimal hyperplasia was markedly increased after balloon injury of the carotid artery in rats, and the main component was VSMCs. PMVs significantly promoted single cell migration and wound closure in vitro. Fluorescence resonance energy transfer revealed that PMVs induced temporal and dynamic calcium oscillations in the cytoplasms of VSMCs. The influx of extracellular calcium, but not calcium from intracellular stores, was involved in the process described above. The channel antagonist GSK219 and specific siRNA revealed that a membrane calcium channel, transient receptor potential vanilloid 4 (TRPV4), participated in the calcium oscillations and VSMC migration induced by PMVs.
Conclusions: TRPV4 participated in the calcium oscillations and VSMC migration induced by PMVs. PMVs and the related molecules might be novel therapeutic targets for vascular remodeling during vascular injury.
 Global reach, higher impact
Global reach, higher impact