Impact Factor
Theranostics 2020; 10(22):9937-9955. doi:10.7150/thno.45488 This issue Cite
Review
Extracellular vesicles in Inflammatory Skin Disorders: from Pathophysiology to Treatment
Department of Dermatology, Xijing hospital, Fourth Military Medical University, Xi'an, Shannxi, 710032, China.
#These authors contributed equally to this work.
Received 2020-2-29; Accepted 2020-7-31; Published 2020-8-7
Abstract
Extracellular vesicles (EVs), naturally secreted by almost all known cell types into extracellular space, can transfer their bioactive cargos of nucleic acids and proteins to recipient cells, mediating cell-cell communication. Thus, they participate in many pathogenic processes including immune regulation, cell proliferation and differentiation, cell death, angiogenesis, among others. Cumulative evidence has shown the important regulatory effects of EVs on the initiation and progression of inflammation, autoimmunity, and cancer. In dermatology, recent studies indicate that EVs play key immunomodulatory roles in inflammatory skin disorders, including psoriasis, atopic dermatitis, lichen planus, bullous pemphigoid, systemic lupus erythematosus, and wound healing. Importantly, EVs can be used as biomarkers of pathophysiological states and/or therapeutic agents, both as carriers of drugs or even as a drug by themselves. In this review, we will summarize current research advances of EVs from different cells and their implications in inflammatory skin disorders, and further discuss their future applications, updated techniques, and challenges in clinical translational medicine.
Keywords: extracellular vesicle, inflammatory skin disorders, biomarker, therapy
Introduction
Extracellular vesicles (EVs) are cell membrane-derived phospholipid bilayer structures that can be produced and released by almost all cell types and range in diameter from 30 to 2000 nm [1]. EVs are found in various biofluids and tissues such as serum, milk, urine, and blister, to name just a few [2]. Currently, EVs are highly heterogeneous and can be classified into three different subtypes based on their size and biogenesis pathway: exosomes (30-150 nm) which are formed by the inward budding of the endosomal membrane during the maturation of multivesicular endosomes, and microvesicles (MVs)/microparticles (100-1500 nm) or apoptotic bodies (500-2000 nm) that are pinched off from the plasma membrane. However, these EV subtypes display overlapping sizes, compositions and densities. Once released into the extracellular space, EVs can be taken up by recipient cells via activation of surface receptors, vesicle internalization (endocytosis), or fusion with target cells to shuttle biological information including various RNAs, lipids and proteins between cells [3]. Thus, EVs regulate multiple biological and pathophysiological processes, including immune responses [4], antibacterial activities [5], cell proliferation and migration [6], and angiogenesis [7, 8], among others, suggesting their pathogenic roles in inflammation progression and cancer metastasis.
Multiple methods have been developed to isolate EVs, including density gradient centrifugation, filtration-based methods and affinity-based or precipitation-based methods. Among them, a density gradient combined with ultracentrifugation is the most widely used and highly recommended method, with low yield and high purity [9]. Regarding identification methods, the International Society of Extracellular Vesicles has released guidelines that include transmission electron microscopy (TEM), NanoSight particle size analysis, dynamic light scattering, and the identification of protein markers by western blotting or flow cytometry [10]. In some studies, researchers used mass spectrometry to analyze EV proteins and quantify their relative abundances or RNA-sequencing (RNA-seq) to analyze the expression of non-coding RNAs, for example, microRNAs (miRNAs), long non-coding RNAs (lncRNAs), or circular RNA (cirRNAs). EV cargoes reflect their cellular origin and surrounding environmental stimuli.
Recent work from us and others has indicated that EVs play key immunomodulatory roles in the pathogenesis of various inflammatory skin diseases, such as psoriasis [11, 12], atopic dermatitis (AD) [13], lichen planus (LP) [14], bullous pemphigoid (BP) [15], systemic lupus erythematosus (SLE) [16], and chronic wound healing [7]. These inflammatory skin disorders pose major problems in dermatology given their complex pathophysiology and refractory nature, which ultimately pose a burden to the health, economic and social systems. As the level of EVs or their cargoes in body fluids may differ between patients and healthy controls, EVs have been used as potential biomarkers of inflammatory skin disorders [17-19]. More importantly, EVs are regarded as ideal therapeutic agents in addition to their native bioactivities, and they can be engineered to deliver a variety of proteins, nucleic acids and/or chemicals or drugs. Here, we discuss the current knowledge on the specificities and regulatory functions of EVs (mainly referring to exosomes and MVs) derived from immune and non-immune cells, their roles in the pathogenesis and treatment of inflammatory skin disorders, and the challenges ahead.
Characteristics and functions of EVs from cells associated with cutaneous inflammation
EVs can be secreted by immune or non-immune cells and affect both innate and adaptive immunity, including antigen presentation, cell differentiation and activation, and immune regulation and suppression, among others. In general, EV surface markers and cargo contents as well as the functions of these EVs are closely related to the pro-inflammatory or anti-inflammatory properties of the parent cells, as summarized in Table 1. Here, we mainly discuss the functions of EVs from cells associated with cutaneous inflammation.
Functions of EVs derived from immune or nonimmune cells
| Source cell | Contents (native) | Contents (modified) | Target cell | Effects | Ref. |
|---|---|---|---|---|---|
| Immune cell-derived EVs | |||||
| DC-EVs | MHC-I and -II, T-cell costimulatory molecules, ICAM-1, MHC-peptide complexes. | DCs | Further initiate the alloreactive T cell responses and acute rejection (in vivo) | [29] | |
| T cells | Promote Th1/Th2 cytokine secretion | [26-28, 32] | |||
| Resident DCs. | Further activate CD4+/CD8+T cells. | [23] | |||
| TLR4 | BMDCs | Increase cellular responsiveness to LPS (in vivo). | [33] | ||
| Circulating antigens and allergens | Mast cells | Induce mast cells to degranulate and trigger anaphylaxis | [34] | ||
| Multiple RNA classes | T cells | Balance Th1/Th2 responses | [24, 25] | ||
| Viral components | T cells or other cells | Transfer infectivity in HCV, Dengue virus, and HIV infection. | [35-37] | ||
| B cell-EVs | MHC-I and -II, costimulatory and adhesion molecules, CD20, CD45, heat shock proteins, and pyruvate kinase type M2. | DCs and others | Promote antigen presentation and T cell responses | [39, 40, 42] | |
| C3 fragments | T cells | [41] | |||
| FasL | Autologous CD4+ T cells | Induce antigen-specific apoptosis | [46] | ||
| Anti-miR-155 | Macrophages | Down-modulate endogenous miRNA | [45] | ||
| T cell-EVs | MHC-I and -II, TCR, CD3, APO2 ligand, adhesion molecules, FasL, and chemokine receptors. | Various | Exert immune regulatory effects, such as inhibiting NK cytotoxicity, regulating DC maturation, and enhancing B cell responses and promoting antibody production. | [4, 49-53] | |
| CD8+T cell-EVs | Mesenchymal tumor stromal cells | Prevent tumor progression | [56] | ||
| MiRNAs or lipids | B cells | Modulate B cell survival, proliferation, and antibody production | [4, 53, 58] | ||
| Th1 cells | Inhibit the proliferation of Th1 cells and IFN-γ secretion | [55] | |||
| MiRNAs | Various | Implicate distinct functions of T cell subsets-derived EVs | [54] | ||
| Not specific | Mast cells | Induce mast cells to degranulate and release several cytokines | [57] | ||
| Macrophage-EVs | Functional proteins | Various | Exert pro-inflammatory role in inflammation | [60-62] | |
| Cholesterol | Lower the cholesterol level | [63] | |||
| Wnt proteins | Dermal papilla cells | Enhance the hair follicle growth | [64] | ||
| MiRs | Monocytes | Induce naïve monocyte differentiation | [65] | ||
| Not specific | Neutrophils | Promote ROS production and subsequent necroptosis | [66] | ||
| Not specific | Fibroblasts, hepatocyte, or epithelial cells, etc. | Contribute to a variety of inflammation and tissue injures, and regulates endothelial cell migration | [67-69, 72] | ||
| Not specific | Human umbilical vein endothelial cells | Inhibit inflammation and accelerate diabetic wound healing (in vivo) | [71] | ||
| Neutrophil-EVs | Neutrophil-associated receptors, granule proteins, annexin A1. | Various | Modulate the pro-inflammatory or anti-inflammatory responses of target cells | [77-84] | |
| Vascular endothelial cells. | Modulate endothelial permeability and vascular remodeling. | [81, 99-104] | |||
| Arachidonic acid | Platelets | Promote platelet-mediated innate immune responses | [87] | ||
| Phosphatidylserine (PS) | Various | Suppress C5a priming of the inflammasome activation (in vivo) | [98] | ||
| Antimicrobial proteins | Bacteria or fungus | Anti-infections | [76, 86] | ||
| Mycobacterium tuberculosis-infected neutrophil-EVs. | Macrophage | Induce autophagy and superoxide anion production in macrophage, thus indirectly promoting the clearance of intracellular mycobacterium tuberculosis. | [88] | ||
| MiRNAs and lncRNAs. | Implicate roles in diagnosis and therapeutics in autoimmunity. | [85] | |||
| Nonimmune cell-derived EVs | |||||
| Mesenchymal stem cell-EVs | Functional proteins | Various | Regulate angiogenesis, apoptosis, inflammation, proteolysis, and extracellular matrix remodeling | [106-112] | |
| Distinct classes of RNAs | |||||
| Pro-angiogenic transcription factors and others | Tubular epithelial cells. | Regulate proliferative or anti-apoptotic pathways | [115] | ||
| Suppress CX3CL1 expression | [116] | ||||
| Promote the recovery of kidney function (in vivo). | [117] | ||||
| Not specific | Various cells in liver diseases. | Suppress the epithelial-to-mesenchymal transition- hepatocyte regeneration | [119] | ||
| Increase hepatocyte regeneration | [119] | ||||
| Decrease proliferation of hepatic stellate cells | [120] | ||||
| Reduce the level of serum alanine aminotransferase and pro-inflammatory cytokines (in vivo). | [121] | ||||
| Not specific | Various | Ameliorate the atopic dermatitis inflammation (in vivo). | [123] | ||
| Keratinocyte-EVs | MHC molecules | T cells | Promote CD4+ and CD8+ T cell proliferation | [126] | |
| Antigens | BMDCs | Help BMDCs to differentiate into mature phenotype and produce large amounts of IL-6, IL-10 and IL-12 | [131] | ||
| β-defensin 2 and chemoattractants | Various | Amplify the pro-inflammatory cascade | [125] | ||
| Functional proteins | Fibroblasts | Modulate wound healing | [128, 129] | ||
| MiRNAs | Help discriminate between EV subpopulations | [127] | |||
| MiR-381-3p | CD4+ T cells | Induce Th1/Th17 polarization in psoriasis. | [164] | ||
| MiR-203 | Melanocytes | Regulate melanin synthesis by melanocytes in skin pigmentation | [130] | ||
| Not specific | Keratinocytes | Suppress cell proliferation | [6] | ||
| Carcinoma cell line TR146 | |||||
| Fibroblast-EVs | Dysregulated collagen-related miRNA | Fibroblasts | Facilitate wound healing | [135] | |
| MiR-21-3p | Cardiomyocytes | Induce cardiomyocyte hypertrophy | [133] | ||
| MiR-23a-3p | Keratinocytes/ epithelial cells | Accelerate cell proliferation and scratch closure | [136] | ||
| Not specific | Fibroblasts | Protect cells against UVB-induced cell death | [137] | ||
| Adipocyte-EVs | Adipokines, Enzymes, immunomodulatory proteins and cytokines, and various mRNAs | Various | Exert regulatory effects on metabolic process and insulin resistance | [143-145, 149, 150] | |
| MiR-27a | Skeletal muscle cells | Induce insulin resistance | [151] | ||
| MiR-155 | Macrophage | Induce M1 macrophage polarization to exacerbate the intestine inflammation (in vivo) | [152, 253] | ||
| MiR-130b-3p | Cardiomyocytes | Exacerbate myocardial ischemia/reperfusion injury | [153] | ||
| Circ_0075932 | Keratinocytes | Promote inflammation and apoptosis | [141] | ||
| Not specific | Vascular cells | Evoke inflammatory responses and vascular remodeling, and increase vascular cell adhesion molecule expression | [154, 155] | ||
| Not specific | Hair-compositing cells | Modulate hair follicle progression | [147] | ||
The contents do not include the common identification markers of EVs;
Abbreviations: BMDCs, bone marrow-derived dendritic cells; DCs: dendritic cells; ICAM-1, intercellular cell adhesion molecule-1; LPS: lipopolysaccharides; MHC: major histocompatibility complex; miR: microRNA; MSCs: mesenchymal stem cells; NK: natural killer; TCR: T cell receptor; Th: T helper; TLR4: Toll-like receptor 4; PD-L1, programmed death-ligand 1.
Immune cell-derived EVs
Dendritic cell (DC)-derived EVs
DCs are the most efficient antigen-presenting cells that take up, process and present antigens to T cells, which contribute to the initiation and progression of immune responses [20]. Early studies reported that DC-derived EVs expressed high levels of functional major histocompatibility complex (MHC) class I (MHC-I) and class II (MHC-II), MHC-peptide complexes, T cell costimulatory molecules, and tumor antigens [21-23]. In addition, it was shown that EVs derived from LPS-induced DCs carried multiple classes of RNAs, including miRNAs, small nucleolar RNAs, Y-RNAs, tRNAs, and small nuclear RNAs [24], which may have roles in balancing T helper 1(Th1)/Th2 responses [25].
DC-EVs have been reported to exert either inhibitory or -stimulatory effects on the immune system, depending on the maturity of the originating cell and the subpopulations of EVs. For instance, EVs from mature DCs carried more MHC-II and costimulatory molecules, which was critical for priming T cell responses and activating the immune system [26-28]. In a murine heart transplant model, donor mature DCs released EVs that transferred MHC molecules to recipient conventional DCs, ultimately initiating alloreactive T cell responses and acute rejection [29]. In contrast, in a rat model of liver transplant, EVs derived from immature DCs induced graft tolerance [30]. Moreover, the antigen presenting capacities of EVs from DCs varied among EV subtypes. For instance, compared to MVs derived from mature DCs, small EVs (sEVs) were much more efficient in inducing antigen-specific CD8+ T cells and eliciting antigen-specific immunoglobulin G (IgG) production [31]. Similarly, sEVs derived from immature DCs promoted the secretion of Th1 cytokines, such as interferon-γ (IFN-γ), while larger EVs (lEVs) induced Th2 cytokine secretion [32]. Other functional proteins, such as toll-like receptor 4 (TLR4), could be transferred between DCs through EVs, which increased cellular responsiveness to lipopolysaccharide (LPS)-induced inflammation in recipient cells [33]. In addition, CD301b+ perivascular DCs continuously released EVs carrying circulating antigens and allergens to neighboring mast cells, and the latter could vigorously degranulate and trigger anaphylaxis [34]. DC-EVs were also responsible for viral evasion and deposition, as DC-EVs may harbor viral components and transfer infection-associated factors in hepatitis C virus, dengue virus, and human immunodeficiency virus infection [35-37].
B cell-derived EVs
B cells are critical modulators of innate and adaptive immune responses as they participate in antigen-specific interactions and antibody production [38]. Similarly, several early studies shown that B cell-EVs expressed a multitude of proteins including MHC-I and MHC-II molecules [39, 40], costimulatory and adhesion molecules, C3 fragments [41], CD20, CD45, heat shock proteins, and pyruvate kinase type M2 [40, 42], which are crucial for antigen presentation and T cell responses. Interestingly, stimulation of receptors on B cells, such as CD40 and IL-4 receptor, or B cell receptor (BCR) cross-linking, could enhance the secretion of EVs [43, 44].
Recently, the functions of B cell-EVs in the tumor microenvironment and inflammation have attracted much attention. EVs derived from CD40/IL-4-induced B cells could be loaded with miRNA-155 mimics or inhibitors using the electroporation method and transferred to macrophage cell lines [45], thus reducing the expression of endogenous miRNAs in recipient cells, which suggested that B cell-EVs are an efficient delivery strategy. Klinker et al. reported that MHC-II+FasL+ EVs isolated from B cell-derived lymphoblastoid cell lines could induce antigen-specific apoptosis in autologous CD4+ T cells, suggesting that this kind of EVs had immunosuppressive functions [46]. Although B cells and derived autoantibodies are crucial for the progression of cutaneous autoimmune diseases, including BP [29, 47] and SLE [48], the roles of B cell-EVs in dermatology are largely unknown and warrant further study.
T cell-derived EVs
T cells are key regulators of the adaptive immune responses involved in inflammatory and autoimmune skin disorders. T cell receptor (TCR) activation and intracellular calcium stimulation could increase EV secretion [49]. It has been shown that T cell-derived EVs contained MHC- I and -II, TCR, CD3, APO2 ligand, adhesion molecules such as CD2 and LFA-1, FasL, and chemokine receptors, among others [49, 50]. These bioactive T cell-EVs could be taken up by different cell types, inducing a variety of immunoregulatory effects, such as inhibiting NK cell cytotoxicity [51], regulating DC maturation [52], and enhancing B cell responses and antibody production [4, 53].
As T cells are classified into several subsets, their EVs exert distinct effects. It was previously reported that EVs released from CD4+T cell subsets, including Th1, Th17 and Treg cells, contained different patterns of miRNAs, suggesting distinct functions for T cell subset-derived EVs [54]. Okoye et al. showed that Treg cell-EVs transferred Let-7d to Th1 cells to inhibit their proliferation and IFN-γ secretion, thus contributing to the suppression and prevention of systemic immune diseases [55]. Activated CD8+ T cells not only prevented tumor progression by direct cytotoxicity against tumor cells but also released EVs to induce mesenchymal tumor stromal cell apoptosis [56]. Moreover, another study revealed that T cell-EVs could stimulate human mast cells to degranulate and release several cytokines, such as IL-24, which in turn activated keratinocytes in vitro [57]. Increasing evidence has suggested that EVs also participated in T-B cognate interactions. T cell-EVs or their delivered miRNAs/lipids regulated B cell survival, proliferation, and antibody production [4, 53, 58], suggesting that T cell-EVs could be engineered to treat B cell overactivation-related diseases.
Macrophage-derived EVs
Macrophages are a highly heterogeneous population that can be activated to differentiate into different phenotypes, including pro-inflammatory M1 or anti-inflammatory M2 macrophages. Macrophages release EVs containing mRNAs that help to identify the phenotype of the parent cells [59]. Using mass spectrometry, the contents of EVs derived from macrophages that were exposed to LPS were identified and included groups of functional proteins, such as plasma membrane-associated proteins, chaperones, metabolic enzymes, cytokines, alarmins, and damage-associated molecular patterns [60-62]. It was also shown that macrophage-EVs were enriched in the active accessible pool of cholesterol, which increased the efficiency of cholesterol transfer to high-density lipoprotein, thus lowering the cholesterol level [63]. A recent study reported that Wnt proteins, such as Wnt3a and Wnt7b, were enriched in macrophage-EVs, thus activating the Wnt/β-catenin signaling pathway to enhance hair follicle growth in vivo and in vitro [64].
On the other hand, macrophage-EVs play a pro-inflammatory role in chronic inflammation and metabolic reprogramming. For instance, macrophage-EVs could induce naive monocyte differentiation via transferring miR-223, which regulated host defense and inflammation [65]. EVs released from hemorrhagic shock-activated macrophage promoted reactive oxygen species (ROS) production in neutrophils and their subsequent necroptosis [66]. In addition, macrophage-EVs activated corresponding recipient cells such as fibroblasts [67], hepatocytes [68], or epithelial cells [69], which contributed to the progression of cardiac injuries, liver injuries, or acute lung injuries. Moreover, once EVs derived from DNA damage-induced macrophages were taken up by recipient cells in vivo, the enhanced cellular glucose uptake and metabolic reprogramming in recipient cells triggered pro-inflammatory responses that augmented chronic inflammation [70]. On the other hand, macrophage-EVs are critical for some anti-inflammatory reactions. For example, in a diabetic skin wound healing rat model, macrophage-EVs inhibited the secretion of pro-inflammatory mediators, thus inhibiting inflammation and accelerating diabetic wound healing [71]. In addition, macrophage-EVs negatively regulated endothelial cell migration by facilitating the internalization and proteolytic degradation of surface integrin β1 [72]. As macrophages are one of the key regulatory cell populations in the context of chronic inflammation [73], the role of macrophage-EVs in psoriasis, AD, and skin regenerating needs to be further elucidated to advance the field.
Neutrophil-derived EVs
Neutrophils are one of the most critical innate immune cells that exert antimicrobial effects via phagocytosis, degranulation, and neutrophil extracellular traps (NETs). Though short-lived, neutrophils can modulate the immune responses in inflammation and cancers [74]. Neutrophil-EVs, first identified by Stein and Luzio [75], were reported to be increased in the circulation during sepsis and inflammation [76]. In response to diverse activators, neutrophils generate EVs with different contents such as neutrophil-associated receptors (CD11b, CD18, CD62L, Fc receptors, and complement receptors) and granule proteins (myeloperoxidase, lactoferrin, elastase, matrix metallopeptidase 9, proteinase 3, heat shock proteins, and S100 calcium-binding protein A8) [77-84]. One recent study showed that 22 miRNAs and 281 lncRNAs were dysregulated in neutrophil-EVs from patients with diffuse cutaneous systemic sclerosis, suggesting a potential role for EVs in the diagnosis and treatment of autoimmune diseases [85].
As neutrophils are indispensable for controlling bacterial and fungal infections, their EVs contribute to the defense against pathogens in several ways. It has been found that neutrophil-EVs were enriched in antimicrobial proteins, which could inhibit the growth and reproduction of bacteria or fungi [76, 86]. Neutrophil-EVs also shuttled arachidonic acid into platelets, and these activated platelets in turn elicited a full neutrophil response, ultimately facilitating neutrophil influx into the lung to eliminate infections [87]. Interestingly, EVs released from Mycobacterium tuberculosis-infected neutrophils could induce autophagy and superoxide anion production in macrophages, thus indirectly promoting the clearance of intracellular Mycobacterium tuberculosis [88].
In addition, neutrophil-EVs can modulate the pro- and/or anti-inflammatory responses of target cells. For instance, neutrophil-EVs enhanced the expression of pro-inflammatory molecules in endothelial cells [89, 90]. Neutrophil-EVs containing neutrophil elastase degraded the extracellular matrix, which exacerbated chronic obstructive pulmonary disease [91]. Moreover, we showed that neutrophil-EVs activated adjacent keratinocytes and increased the expression and production of pro-inflammatory mediators, which induced a vicious cycle in severe pustular psoriasis [82]. In addition, neutrophil-EVs exerted anti-inflammatory effects, such as reducing pro-inflammatory mediators to protect the cartilage [92], inhibiting the production of pro-inflammatory cytokines and enhancing anti-inflammatory cytokines in NK cells [93], monocyte-derived DCs [94], or macrophages [95-97], which indirectly limited excessive inflammatory responses. Neutrophil-EVs from joint aspirates of gouty arthritis patients had similar anti-inflammatory properties. In response to C5a, neutrophils released phosphatidylserine-positive EVs to suppress the inflammasome activation that was primed by C5a and consequently inhibited IL-1β release and neutrophil influx [98].
Furthermore, neutrophil-EVs also modulate endothelial permeability and vascular remodeling in a cargo-dependent manner. Neutrophil-EVs were able to disrupt junctional integrity and increase permeability due to the activities of S100A8, S100A9, myeloperoxidase (MPO), and cathepsin G, among others [81, 99, 100-102]. In contrast, neutrophil-EVs transferring barrier-protecting proteins, such as annexin 1, maintained junctional integrity and decreased permeability [103, 104]. Thus, it is worth further exploring the regulatory role of neutrophil-EVs in inflammatory skin diseases that display vascular remodeling in skin lesions.
Non-immune cell-derived EVs
Mesenchymal stem cell-derived EVs
Mesenchymal stem/stromal cells (MSCs) are self-renewing, multipotent stromal cells that can differentiate into a variety of cell types. MSCs mainly exert immunomodulatory effects associated with tissue homeostasis and regeneration. MSC-derived EVs are reported to have multiple biological functions, including anti-inflammation, tissue repair, immunosuppression, and neuroprotection [105]. Thus, recent studies have focused on exploiting MSCs-EVs as a possible noncellular therapy, which will be discussed in Section 4 of this review. RNA-seq studies showed that MSC-EVs were enriched for distinct classes of RNAs [106-108], and the proteomics profiling of MSC-EVs identified functional proteins involved in cell proliferation and apoptosis, inflammation, extracellular matrix remodeling, and angiogenesis [109-112].
In general, MSC-EVs mainly play protective roles in inflammation. For instance, in tubular or renal injury, MSC-EVs regulated proliferative or anti-apoptotic pathways in tubular epithelial cells [113, 114], shuttled several pro-angiogenic transcription factors [115], or suppressed CX3CL1 expression and reduced the subsequent infiltration of macrophages in the damaged kidney [116]. Recent in vivo studies further indicated that MSC-EVs promoted the recovery of kidney function in animal models of ischemia-reperfusion-induced acute kidney injury, which could be considered as a future potential therapy [117]. Moreover, some studies have reported that MSC-EVs could be used for the treatment of liver diseases, as MSC-EVs suppressed the epithelial-to-mesenchymal transition [118], increased hepatocyte regeneration [119], and decreased the proliferation of hepatic stellate cells [120]. They also reduced the level of serum alanine aminotransferase and pro-inflammatory cytokines in liver injury mice, suggesting that MSC-EVs have anti-inflammatory effects in liver injury [121]. Human umbilical cord MSC-EVs carrying circular RNAs could inhibit ischemia-induced pyroptosis and the release of downstream IL-1β and IL-18, which helped repair ischemic muscle injury [122].
In dermatology, adipose tissue-derived MSC-EVs attenuated pathological symptoms in an AD mouse model, reducing clinical scores levels of IgE and eosinophils in the blood, the infiltration of immune cells in skin lesions, and the mRNA expression of various inflammatory cytokines [123], which indicated that MSC-EVs could be a novel and promising therapeutic strategy for AD treatment.
Keratinocyte-derived EVs
Keratinocytes, the main components of the epidermis, can sense pathogens and mediate immune responses. Dysregulation of keratinocytes and their crosstalk with other immune cells gives rise to the initiation and propagation of inflammatory skin diseases in susceptible individuals [124]. Similar to EVs derived from other cells, keratinocyte-EVs also vary in composition and abundance of contents depending on the parent cell status and stimulus. For example, IL-17A-treated keratinocytes released EVs containing β-defensin 2 and chemoattractants such as CXCL1, CXCL3, CXCL5, and CXCL6 [125]. In addition, Staphylococcus aureus (S. aureus) enterotoxin B-loaded HaCaT cells (a keratinocyte cell line) released EVs containing MHC molecules, which promoted CD4+ and CD8+ T cell proliferation in vitro [126]. Keratinocyte-EVs were also reported to carry a set of miRNAs that helped discriminate between EV subpopulations [127].
Keratinocytes-EVs are actively involved in cellular cross-talk, thus regulating various functions associated with skin homeostasis, including wound healing, proliferation, and pigment production. For example, keratinocyte-EVs carried cathepsin B, transforming growth factor binding protein, and matrix metalloproteinase-1, which stimulated fibroblasts to facilitate extracellular matrix remodeling and subsequent keratinocyte migration during wound healing [128, 129]. In skin pigmentation, EVs from ultraviolet B-irradiated keratinocytes significantly increased both the expression and activity of melanosomal proteins in melanocytes [130], partially explaining how the cell crosstalk regulated pigmentation. In addition, EVs derived from keratinocyte induced by IFN-γ could be internalized by bone marrow-derived cells, and the latter cells differentiated into a mature phenotype with enhanced CD40 expression and increased IL-6, IL-10, and IL-12 production [131]. Given that keratinocytes play vital roles in host defense and immune responses in skin lesions, it is hypothesized that their EVs may act as regulators in the complicated immune disorders of inflammatory skin diseases.
Fibroblast-derived EVs
Fibroblasts can synthesize the major components of the extracellular matrix in connective tissue, thus maintaining the structural integrity of most tissues. Increasing evidence indicated that fibroblasts and their derived EVs exhibited functional specializations according to their source organ and spatial location [132]. For instance, cardiac fibroblasts-EVs could transfer miR-21-3p to cardiomyocytes and induce cardiomyocyte hypertrophy [133]. Fibroblast-EVs from patients with eosinophilic asthma promoted epithelial cell proliferation, thus contributing to airway remodeling in severe asthma [134]. Fibroblast-EVs from scleroderma patients exhibited dysregulated collagen-related miRNA levels and further upregulated the expression of type I collagen in fibroblasts to facilitate wound healing [135]. Similarly, fibroblast-EVs could shuttle miR-23a-3p to accelerate scratch closure of epidermal keratinocytes in vitro [136]. Moreover, fibroblast-EVs significantly inhibited the production of ROS and cell death in fibroblasts induced by ultraviolet B radiation, thus playing a critical role in skin homeostasis during photoaging [137].
Adipocyte-derived EVs
Nowadays, the adipose tissue has been recognized as an endocrine organ that secretes pleiotropic bioactive molecules that modulate metabolism in distant organs and immune cell functions [138]. Adipogenesis was thought to affect EV structure, molecular composition, and function, as EV production was higher in cells before adipogenesis [139]. Interestingly, mouse perigonadal adipose tissue in leptin-deficient obese mice released more EVs than that of lean mice [140], and the cargoes and regulatory functions of these EVs were also different [141, 142]. Protein profiling has revealed that EVs released by adipose tissue carried adipokines such as adiponectin, IL-6, monocyte chemoattractant protein-1, and resistin [143]. In addition, these EVs also harbored enzymes including fatty acid synthase, acetyl-CoA carboxylase, glucose-6-phosphate dehydrogenase, immunomodulatory proteins and cytokines, as well as various mRNAs [144, 145]. Further, two subpopulations of adipocyte-EVs, small EVs and large EVs, were identified and shown to exhibit specific protein signatures [146]. Notably, the functions of EVs released from adipocytes in different differentiation states were varied. For instance, EVs from immature adipocytes were shown to induce telogen-to-anagen transitions in hair follicles, whereas those from mature adipocytes inhibited hair follicle progression [147]. Therefore, it can be hypothesized that under different circumstances, adipocytes secrete EVs with distinct regulatory functions, contributing to diverse immune responses.
Adipocyte-EVs, now regarded as a new adipokine, exert regulatory functions in metabolic processes and insulin resistance [148]. It was reported that circulating EVs were significantly increased in obese mice and humans compared to lean controls, and could be biomarkers of metabolic stress [149]. Besides, EVs derived from the adipocytes of obese mice increased appetite and weight when administered to lean mice, whereas EVs from the adipocytes of lean mice decreased the weights of obese mice [150], indicating their regulatory role in obesity and metabolic diseases. Another study reported that obese adipocytes released high levels of exosomal miR-27a, which repressed PPAR-γ and induced insulin resistance in skeletal muscle [151].
In addition, adipocyte-EVs play pathogenic roles in immune responses via exerting effects on various cells. Adipocyte-EVs from obese mice induced M1 macrophage polarization to exacerbate intestinal inflammation [152]. In addition, EVs from diabetic adipocytes or high glucose/high lipid-challenged non-diabetic adipocytes were enriched in miR-130b-3p, which aggravated myocardial ischemia/reperfusion injury in the diabetic heart [153]. Similar findings have been reported in perivascular adipose tissues, where adipocyte-EVs from obese mice evoked inflammatory responses and vascular remodeling [154]. Moreover, TNF-α/TNF-α+hypoxia-induced adipocyte-EVs enhanced neutrophil adhesion by increasing vascular cell adhesion molecule expression on one vascular cell line [155]. Considering the critical roles of adipocytes in skin repair and inflammatory states, future studies of these EVs will have far-reaching implications.
EV involvement in the pathophysiology of inflammatory skin disorders
Chronic inflammatory skin diseases, including psoriasis, AD, LP, and SLE, among others, are refractory with long-lasting courses. Though biological drugs such as anti-TNF, anti-IL-17, and anti-IL-12/23 agents show treatment-associated benefits, the complex pathogenesis is insufficiently understood, which warrants further exploration. Here, we will mainly describe how EVs participate in complicated pathophysiological processes involved in inflammatory skin disorders.
Psoriasis
Psoriasis is the most common chronic inflammatory skin disease and is characterized by abnormal proliferation and differentiation of keratinocytes and massive infiltration of immune cells [156]. A number of studies have shown that endothelial cell- and platelet-derived EVs were increased in patients with psoriasis [157-161], were positively correlated with the psoriasis area and severity index score [157], and were decreased by anti-TNF-αbut not anti-IL-12/23 treatment [162]. Besides, psoriasis-related cytokines modulated the production of EVs, as IL-17A induced HaCaT cells to produce EVs carrying the mRNAs of several chemokines and β‐defensin 2 [125]. Recent studies attempted to analyze the miRNA profiles in plasma-derived EVs to illustrate their potential as future psoriasis biomarkers [17, 163].
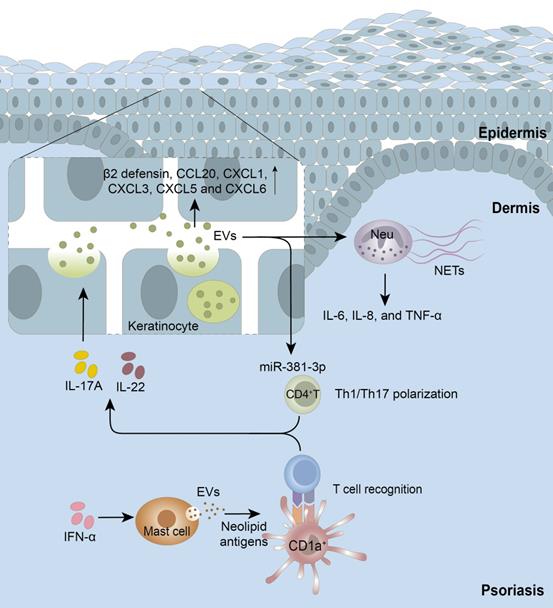
Studies on the functions of EVs in psoriasis have gradually developed in recent years (Figure 1). As one early study revealed, IFN-α induced mast cells to release EVs that were capable of transferring cytoplasmic PLA2 activity to neighboring CD1a-expressing cells, which further led to the generation of neolipid antigens and subsequent recognition by CD1a-reactive T cells [11], which established EVs as critical mediators in psoriasis. Our recent study demonstrated that EVs isolated from psoriatic cytokine-induced keratinocytes could be endocytosed by neutrophils and induced the latter to produce NETs and pro-inflammatory cytokines, thus exacerbating psoriatic inflammation [12]. Interestingly, neutrophils from patients with generalized pustular psoriasis secreted more EVs than those from controls, and further triggered keratinocytes to produce high levels of inflammatory molecules, such as IL-1β, IL-36G, IL-18 and TNF-α [82]. These results suggest that EVs are critical mediators of keratinocyte-neutrophil crosstalk in the pathogenesis of psoriasis. Moreover, our latest study revealed that EVs derived from psoriatic keratinocytes transferred miR-381-3p to CD4+ T cells, inducing Th1/Th17 polarization and promoting psoriasis development [164]. Interestingly, it was reported that EVs from two similar diseases, rheumatoid arthritis (RA) and psoriatic arthritis (PsA), showed divergent effects, as RA-derived or healthy control-derived EVs profoundly inhibited osteoclast differentiation while PsA-derived EVs had a stimulatory effect [165]. Therefore, by comprehensively studying the biological functions of EVs, we will gain important insights regarding their roles in the immune network of psoriasis, or other associated syndromes.
Extracellular vesicles (EVs) in the cell-cell crosstalk of psoriasis. Keratinocyte-EVs induced by psoriatic cytokines such as IL-17A can be taken up by neighboring keratinocytes to upregulate mRNA expression of β2 defensin and chemokines, by neutrophils to induce NETosis and the production of IL-6, IL-8, and TNF-α in neutrophils, or by CD4+T cells to facilitate Th1/Th17 polarization. In turn, neutrophil-EVs also induce keratinocytes to produce a variety of chemokines to attract more immune cells. In addition, IFN-α promotes mast cells to secrete EVs, which induces the generation of neolipid antigens and subsequent recognition by lipid-specific CD1a-reactive T cells. Neu, neutrophils; NETs, neutrophil extracellular traps.
Atopic dermatitis
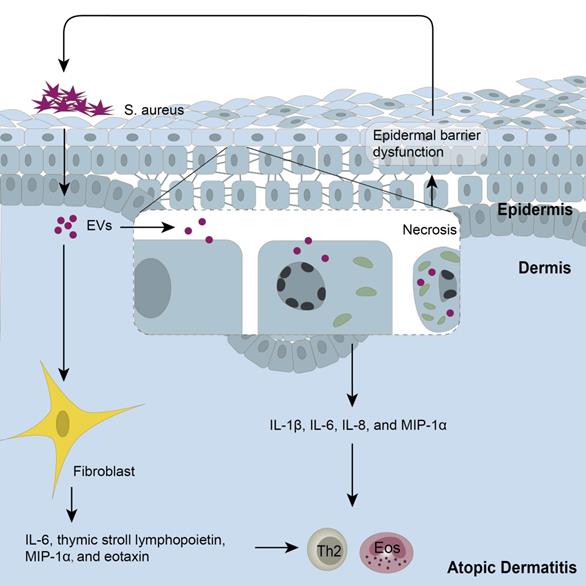
AD is another common inflammatory skin disease, characterized by typical type 2 skin inflammation with a defective barrier [166, 167]. As AD patients are susceptible to S. aureus infection, which in turn aggravates AD inflammation-associated, several studies have explored the role of S. aureus-derived EVs in AD. S. aureus-EVs could exacerbate AD inflammation by delivering bacterial effector molecules to host cells, thus aggravating the inflammatory responses (Figure 2). For instance, it was reported that S. aureus-EVs efficiently increased the production of pro-inflammatory mediators such as IL-6, thymic stromal lymphopoietin, and macrophage inflammatory protein-1α in dermal fibroblasts [168], triggered HaCaT cells to overexpress pro-inflammatory cytokines including IL-1β, IL-6, IL-8, and MIP-1α [169], and induced endothelial cell activation and monocyte recruitment [13]. In addition, it was shown that α-hemolysin transported in S. aureus-EVs induced keratinocyte necrosis, exacerbating both skin barrier disruption and AD-like skin inflammation [170]. Thus, S. aureus-EVs may be regarded as one of the therapeutic targets for the management of AD aggravation. Notably, EVs derived from thymol-treated S. aureus or Lactobacillus plantarum alleviated the AD-like skin lesions including epidermal thickening and IL-4 level [171, 172], indicating their potential to treat AD.
Extracellular vesicles (EVs) in the pathogenesis of AD. On the one hand, EVs secreted from S. aureus could induce keratinocyte necrosis and damage to epidermal barrier structures and functions, which in turn facilitates further colonization of S. aureus-EVs. These keratinocytes produce IL-1β, IL-6, IL-8, and MIP-1α in response to S. aureus-EVs. On the other hand, S. aureus-EVs upregulate the production of pro-inflammatory mediators in fibroblasts, including IL-6, thymic stromal lymphopoietin, macrophage inflammatory protein-1α, and eotaxin, contributing to the Th2 immune response in AD pathogenesis. S. aureus, Staphylococcus aureus.
Lichen planus
LP is the third most common inflammatory skin disease, characterized by epidermal keratinocyte death and dense infiltration of T cells in the dermis [173]. We previously provided evidence of the role of IFN-γ in the cell-mediated cytotoxicity of keratinocytes in LP [174]. Recently, several studies explored the function of EVs in LP. For instance, one study profiled salivary EVs from patients with oral LP and identified miR-4484, miR-1246, and miR-1290 as enriched miRNAs in LP, making these molecules potential biomarkers for oral LP [19]. In addition, it was shown that circulating EVs in erosive oral LP patients enhanced T cell proliferation and attenuated T cell apoptosis [14]. Thus, further studies are warranted to profile and determine the pathogenic roles of EVs derived from skin lesions or circulation of LP patients.
Bullous pemphigoid
BP is a severe autoimmune inflammatory disorder and clinically manifests as subepidermal blisters and erosions of the skin and/or mucous membranes [175]. Blister fluid contains infiltrated immune cells, cytokines, and chemokines, as well as EVs, all of which are capable of exerting biological functions and promoting inflammatory responses. We showed that EVs isolated from the blister fluid of BP patients could be internalized by human keratinocytes, which led to the production of critical inflammatory cytokines and chemokines, enhancing neutrophil trafficking to skin lesions and triggering local autoinflammatory responses [15]. Neutrophils were reported to be critical for blister formation and exacerbated inflammation in BP [176, 177]. We also employed mass spectrometry to elucidate the proteome of blister fluid-derived EVs for the first time, showing that they contained a variety of proteins implicated in autoimmunity and inflammatory responses [15]. However, the contributions of neutrophil-, T cell-, B cell-, and other monocyte-EVs to the initiation and progression of BP are largely unknown.
Systemic lupus erythematosus
SLE is a chronic, refractory, and systemic autoimmune disease characterized by circulating autoantibodies and the formation of immune complexes. It harms multiple organs and presents a variety of clinical manifestations [178]. To date, the contents and functions of EVs carrying autoantigens, cytokines, surface receptors, and non-coding RNAs have been studied in SLE.
As early studies reported, the total number of EVs and IgG-positive EVs were increased in the plasma of SLE patients [18, 179], and their high levels were positively correlated with anti-DNA levels, suggesting that these EVs could represent an important source of immune complexes in SLE [180]. Nielsen et al. showed that the level of annexin V non-binding cell-derived EVs was positively correlated with disease severity and some systemic indicators [181]. They further found that the levels of IgG, IgM, and C1q were elevated in EVs from SLE patients compared with those from healthy controls [182]. Nevertheless, it was not clearly demonstrated whether IgG in EVs had autoantibody activity. It was also reported that CD31+/annexin V+/CD42b- MVs [182] or CD41+ EVs harboring IgG [183] were higher in SLE patients than in controls. Using high-sensitivity nano-liquid chromatography tandem mass spectrometry, 248 proteins were found to be significantly upregulated in the circulating EVs isolated from SLE patients relative to those of the controls [184]. In addition, another recent study measured EVs by flow cytometry to identify small (0.2-0.7 μm) and large (0.7-3.0 μm) EVs, and showed that patients with active lupus nephritis had increased levels of large EVs containing mitochondria (mitoEVs) and IgG-positive mitoEVs, indicating that distinct EV subpopulations can have different functions in SLE [185]. Further, urinary EVs of SLE patients were also identified and enriched in miRNAs, with miR-146a being the most prominent [186].
Moreover, it was shown that these increased EVs in the plasma of active SLE patients induced ROS production and degranulation in neutrophils [187], activated pDCs to secrete IFN-α via TLR7 [188], or contributed to MSC senescence in SLE [189]. The elevated EVs and their immune complexes in SLE patients also promoted secondary leukocyte infiltration by regulating vascular remodeling and chemokine secretion [16]. Moreover, EVs derived from platelets mostly harbored IgG and overexpressed CD69 and CD64, and promoted pro-inflammatory responses in monocytes, thus exacerbating SLE-associated inflammation [190]. Thus, it is worth noting that EVs play a crucial role in initiating and aggregating autoimmune reactions in SLE.
Chronic wound healing
Wound healing is a complex and dynamic process that includes hemostasis, inflammation, angiogenesis, re-epithelialization and remodeling [191]. However, the disturbances in this process, common in diabetes and aging individuals, lead to chronic wounds, a dilemma in dermatology [192, 193]. A variety of cell types with distinct roles are involved in the different phases of chronic wound healing, including neutrophils [194], macrophages [195], mast cells [196], DCs [197], T cells [198], fibroblasts [199], and others [192]. Therefore, EVs, cytokines, growth factors, and chemokines derived from multiple sources can actively regulate complex cellular signaling networks.
Numerous studies have focused on pathogenic factors and new techniques to promote wound healing. According to recent studies, EVs can contribute to multiple stages of wound healing cascades such as cell proliferation and differentiation [200], coagulation [201], angiogenesis [202], and extracellular matrix remodeling [203]. For instance, EVs isolated from platelet-rich plasma could effectively induce the proliferation and migration of endothelial cells and fibroblasts to improve angiogenesis and re-epithelialization in chronic wounds, therefore showing efficacy in chronic wounds in a diabetic rat model [204]. EVs released from corneal epithelial cells were enriched in provisional matrix proteins, fibronectin, and thrombospondin-1, promoting the differentiation of myofibroblasts in the development of corneal scars [200]. Similarly, EVs carrying miR-21, which were mainly derived from resident keratinocytes, were elevated in the wound fluid of healing chronic wound patients, and able to convert M1-polarized human macrophages to fibroblast-like cells; however, the conversion was strikingly impaired in a mouse model of experimental diabetes, and could be rescued by nanoparticles delivering miR-21 to macrophages [205]. However, advanced glycation end products induced human umbilical vein endothelial cells to secrete EVs enriched in miR-106b-5p, which triggered fibroblast autophagy, thus decreasing collagen synthesis and delaying cutaneous wound healing [206]. EVs derived from normal and diabetic human corneolimbal keratocytes exhibited distinct contents, and accelerated or delayed the proliferation of limbal epithelial cells in wound healing [207]. Therefore, we hypothesize that EVs derived from various sources contribute to the local immune responses in chronic wound healing.
Therapeutic approaches of EVs in inflammatory skin disorders
EVs as biomarkers in inflammatory skin disorders
EVs are mainly regarded as diagnostic and prognostic biomarkers in cancer but are still largely unexplored in skin inflammatory diseases. Recently, increasing evidence has indicated their potential role as noninvasive biomarkers for predicting the onset, relapse or reaction to drugs in the field of inflammatory skin disorders.
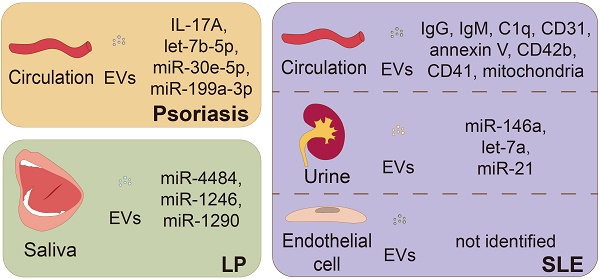
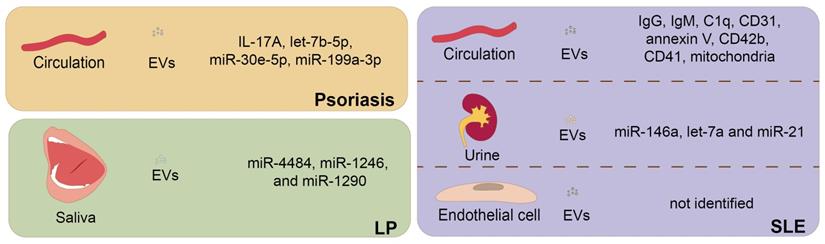
Several studies on psoriasis, SLE, AD, and LP [19] aimed to explore the role of EVs as biomarkers (Figure 3). For instance, the level of IL-17A+ EVs in circulation was significantly higher in patients with moderate-to-severe psoriasis than in those with mild psoriasis, suggesting that the components of EVs could be indicators of distinct disease stages [161]. Another study further compared the plasma-derived EV miRNAs from cutaneous-only psoriasis patients (n=15) with those of PsA patients (n=14), showing that let-7b-5p and miR-30e-5p in plasma-derived EVs were significantly lower in PsA patients, which suggested that circulating EV miRNAs might serve as biomarkers for PsA [163]. A recent study profiled the miRNAs of EVs from psoriasis patients (n=52) and healthy controls (n=26) and revealed that 26 miRNAs were upregulated and 24 were downregulated, with miR-199a-3p the most differentially expressed in psoriasis [17]. These results highlight the potential of EVs as diagnostic markers for psoriasis patients, however, larger samples and the pathogenic roles of EVs should be considered in the future. In SLE, several studies have suggested that the levels of circulating EVs were correlated with disease activity and clinical features, indicating that EVs could be reliable biomarkers of SLE activity [18, 181, 184]. For instance, it was shown that the increase in endothelial EVs was positively correlated with disease activity, glomerulonephritis, and vascular dysfunction [181], and was reduced by immunosuppressive therapy with decreasing cardiovascular risk [208]. Urinary EVs containing let-7a and miR-21 were significantly down-regulated in lupus nephritis patients and elevated after complete course of effective treatment, suggesting that urinary EV-associated miRNAs could be used as liquid biopsies to estimate the clinical stage of lupus nephritis patients [209]. In addition, as one recent study preliminarily explored, serum microbial EVs showed potential as novel biomarkers for AD diagnosis [210].
Extracellular vesicles (EVs) are potential biomarkers in inflammatory skin disorders. EVs derived from various origins have the potential to serve as biomarkers in several inflammatory dermatoses, including psoriasis, LP, and SLE. In psoriasis, the level of IL-17A+ EVs in circulation was significantly higher in patients with moderate-to-severe psoriasis than in those with mild psoriasis. MiR-199a-3p was the most up-regulated in serum EVs from psoriasis patients, implicating its diagnostic role in future study. Let-7b-5p and miR-30e-5p in serum EVs were significantly lower in patients with PsA, suggesting that circulating EV miRNAs might serve as biomarkers for PsA. In SLE, the increase in endothelial EVs or circulating EVs carrying several immune molecules was positively correlated with disease activity. And urinary EVs of SLE patients were also identified and enriched in miRNAs, with miR-146a being the most prominent. Urinary EVs containing let-7a and miR-21 were significantly down-regulated in lupus nephritis patients, and elevated after the complete course of effective treatment. In LP, salivary EVs of patients from oral LP were enriched in miR-4484, miR-1246, and miR-1290, making them potential biomarkers for oral LP.
Further studies of the novel ways that EVs can reflect disease pathogenesis or clinical stages, predict relapse or prognosis, indicate responses to therapies, or guide therapy beyond current biomarkers are needed.
EVs as therapeutic agents in inflammatory skin disorders
Currently, EVs are being explored as nanotherapeutic agents for immune therapy, regenerative medicine, and drug delivery. In dermatology, studies on EVs are just beginning to show promising prospects in inflammatory skin disorders. As mentioned above, adipose tissue-derived MSC-EVs showed promising results as a cell-free therapeutic modality for AD treatment [123]. Several other investigations have demonstrated the potential uses of MSC- and APC-derived EVs as cell-free agents to treat autoimmune diseases. It was reported that the transplantation of MSCs would rescue bone marrow MSC function in a lupus knock-out mouse model via transferring EVs-derived Fas to recipient cells [211]. Another study showed that MSC-EVs were successfully used to treat a patient with refractory graft-versus-host disease and showed sustained alleviation of cutaneous and mucosal manifestations after 4 months [212].
Moreover, several studies have shown that EVs can be used as therapeutic agents in wound healing. EVs derived from umbilical cord blood were shown to be enriched in miR-21-3p, and promoted the proliferation and migration of fibroblasts and enhanced the angiogenic activities of endothelial cells, thus accelerating re-epithelialization and cutaneous wound healing [213]. Keratinocyte-EVs could also modulate fibroblast function and angiogenesis by transferring miR-21 to facilitate skin wound healing [7]. EVs from oral mucosa epithelial cells of human healthy donors showed pro-regenerative effects on skin wound healing [214], and EVs from human urine-derived stem cells could promote angiogenesis and wound healing in diabetic mice [215]. Interestingly, Synechococcus elongatus-EVs were capable of augmenting endothelial angiogenesis and stimulating new blood vessel formation, indicating that Synechococcus elongatus-EVs maybe a promising strategy for wound healing [202]. It was shown that MSCs-EVs maintained or accelerated rapid wound healing by activating fibroblasts [203], epithelial cells [216], or gingiva wound healing [217], which provided a potential therapeutic approach in this field.
Nowadays, the combination of EVs and biomaterials to enhance the wound healing process represents a novel approach for chronic wound therapy [218]. One study developed an injectable antibacterial hydrogel with stimuli-responsive adipose-derived MSC-EVs, which could efficiently accelerate chronic wound healing and skin regeneration with coordinated actions [219]. More recently, a study reported that a light-activated hydrogel containing small EVs isolated from human mononuclear cells promoted wound healing, a process that was partially orchestrated by EV-miRNAs [220]. Additionally, allogenic EVs isolated from adipose tissue-derived stromal cells were engineered into a thermoresponsive gel, which resulted in a statistically significant improvement in fistula healing [221]. To date, several phase-I or phase-II clinical trials have been conducted to study whether EVs can be used in humans without life-threatening complications [222, 223]. However, only one clinical trial was established to study the use of EVs in the treatment of skin diseases. AGLE-102 is an allogeneic EV derived from normal donor MSCs. One phase 1/2A, multi-center, randomized study aimed to assess the safety and efficacy of AGLE-102 in the treatment of lesions in epidermolysis bullosa patients (NCT04173650).
Importantly, EVs, especially exosomes, have great potential as drug delivery vehicles. They are suitable for delivering therapeutic agents due to their natural properties, including material transportation, stability in circulation, relatively long half-lives, and excellent biocompatibility [224]. For instance, EV-based drug formulations offer a powerful and novel delivery platform for anti-cancer and -inflammation therapy [225-227]. For example, since the transcription factor nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) plays a pivotal role in modulating the inflammatory cascades in a variety of inflammatory diseases, including septic shock and psoriasis, an optogenetically engineered EV system was employed to load a large amount of super-repressor IκB into EVs. These engineered EVs were taken up by recipient cells and then attenuated systemic inflammation in septic model mice [228]. Though several techniques are in use for drug loading and targeted delivery, a standard effective method is still lacking. Existing methodologies include the incubation of drugs with EVs, electroporation or saponification to induce the formation of small pores within the membranes, freeze-thaw methods that can cause the degradation of many EV proteins, and the transfection of nucleic acids into the secreting cells, among others [229]. Any method can be selected depending on the study demand and its advantages. Therefore, to exploit the therapeutic potential of EVs, innovative approaches and full considerations are required.
Based on the availability, cost of production and safety, plants, fruits, and even milk can be used as sources from which to isolate EVs for clinical use [230]. For instance, anti-tumor drugs such as doxorubicin and curcumin were loaded on grapefruit-EVs decorated with targeting modifications, and showed anti-inflammatory effects in tumor-bearing mice [231]. Milk-EVs were reported to survive harsh and degrading conditions in the gut and then taken up by various cell types. Thus, several studies isolated EVs from milk and attempted to load them with various drugs with high yield and safety [232-234]. Thus, these innovations and updated techniques can be considered in the future study of EV utility in inflammatory skin diseases.
In dermatology, the combination of EVs and biomaterials in regenerative medicine warrants in-depth study in mouse models and clinical trials, for instance, in wound healing and hair regeneration. The topical use of this kind of EV will also show promising prospects in treating inflammatory skin diseases. With improved understanding of EV functions and the development of related bioengineering strategies, it is possible to develop therapeutic applications such as EVs enriched in the cargo of interest, with enhanced targeting properties or decorated with physiological or synthetic ligands to target specific receptors that are overexpressed in different autoimmune diseases.
Challenges and Future Directions
Currently, several major challenges limit the broader translational use of EVs. As the EV classifications are continuously evolving, one problem is the lack of unique markers for the heterogeneous subclasses of EVs, which overlap in sizes, biophysical properties, and contents [235]. Therefore, the ISEV suggested the use of physical characteristics (such as small EVs), biological composition (some protein positive EVs), or descriptions of conditions (such as hypoxic EVs) to name the EV subtypes [236]. Since the isolation methods and approaches for EV analysis are evolving [237], standard methods need further validation. One proposed solution is to comprehensively study the biogenesis and cargo content of all EVs from multiple cell types [238]. Using asymmetric flow field-flow fractionation, one group identified two exosome subpopulations, large exosomes (90-120 nm) and small exosomes (60-80 nm), and discovered an abundant population of non-membranous nanoparticles termed “exomeres” (~35 nm). Each subpopulation showed unique protein and nucleic acid profiles, as well as biophysical properties, suggesting a distinct biological function for each subpopulation [239]. Besides, fourier-transform infrared spectroscopy provided collective fingerprints of EV subpopulations including large (~600 nm), medium (~200 nm), and small (~60 nm) EVs [240]. Moreover, diverse subpopulations of plasma EVs were identified using high-sensitivity fluorescence-activated vesicle sorting and showed that erythrocyte- and platelet-derived EVs carried distinct repertoires of nucleic acids that were similar to those of their original sources [241]. Therefore, as increasing evidence has shown that EV subclasses carry different cargoes, it is necessary to develop standardized and effective methods to identify and isolate the different EV subpopulations.
Current EV isolation methods yield many non-vesicular proteins and contaminated EVs, which limit the profiling of nucleic acids or proteins in EVs. Thus, several techniques were developed to improve our understanding of EV contents. A recent study used a trypsin-digested proteomics approach to classify trypsin-sensitive and trypsin-resistant vesicular proteins, and to systematically study the actual-vesicular proteins, which helped to identify reliable functional proteins, and revealed their pathophysiological roles, an important aspect in the use of EVs as liquid biopsies [242]. In addition, new techniques such as exosome-templated nanoplasmonics [243] and nanopatterned microchips [244] were recently developed to accurately and sensitively monitor the molecular profiles of EVs, which would facilitate the development of liquid biopsies.
Further, how to identify and clearly observe EVs has been a major obstacle in understanding EVs and their utility. Notably, new technologies have flourished in recent years. For instance, one study used a non-pH-sensitive red fluorescent tag to visualize the lifecycle and biogenesis of EVs, including multivesicular body (MVB) trafficking, MVB fusion, EV uptake, and endosome acidification [245]. To quantify EV uptake at the single-cell level, another study developed an engineering approach that combined mass cytometry with highly multivariate cellular phenotyping. This innovation could help to unravel the in vivo fate of EVs taken up by recipient cells, elucidating the mechanism of action of EVs in vivo [246]. The updated technologies tracking EVs will improve our mechanistic understanding of the biodistribution of EVs.
Nowadays, efforts to increase the production of EVs by cells for future therapeutic applications are ongoing. Conventional isolation methods such as ultracentrifugation require multiple steps that cause significant loss and damages to EVs. To overcome this limitation, researchers have developed several methods to increase the yield of EVs or to engineer EVs. One strategy is to increase EV biogenesis in the donor cell by overexpressing regulatory proteins involved in EV biogenesis [247] or increasing MVB docking at the plasma membrane [248]. Besides, physical conditions including radiation, hypoxia, and low pH were reported to upregulate EV production. For instance, nitrogen cavitation was used to instantly disrupt neutrophil-like HL60 cells leading to a 16-fold increase in the formation of nanosized membrane vesicles, which were then loaded with an anti-inflammation drug to treat acute lung inflammation/injury and sepsis induced by LPS [249]. Ultrasonication of MSCs for 1 min also improved the EV yield, which exhibited ∼20-fold higher and ∼100-fold faster production than the natural secretion [250]. A recent study described a detailed method to fabricate an origami-paper-based device that integrated the ion concentration polarization technique for effective enrichment and isolation of EVs in a simple manner [251]. Further, engineered EVs showed promising roles in this field. One group developed a system to engineer EVs to sustainably release TGF-β1, IL-2, and rapamycin to induce Treg differentiation from naïve T cells [252]. However, more studies should be conducted to ensure that the engineered EVs exhibit stable bioactivities and efficacies, stable packing of biological materials, and little cytotoxicity.
Conclusions
In recent years, we have witnessed a boom in EV studies in the context of nearly all diseases, such as in the dysregulation and treatment of cancers, inflammation, and autoimmune diseases. Therefore, we reviewed the regulatory functions of immune and non-immune cell-derived EVs, and their roles in inflammatory skin disorders, as triggers of immune responses, potential biomarkers, or therapeutic agents. However, the studies are just beginning in dermatology, and the precise contents of most EV preparations or their multiple functions remain to be deciphered. Likewise, new strategies and more comprehensive studies are needed to identify EV subpopulations with high accuracy and selectivity, and to address the emerging challenges. We are optimistic that continuing studies on EVs will allow future application of EVs in the detection and treatment of various inflammatory skin diseases.
Abbreviations
AD: atopic dermatitis; APCs: antigen presenting cells; BCR: B cell receptor; BP: bullous pemphigoid; DCs: dendritic cells; ECM: extracellular matrix; EV: extracellular vesicle; IFN-γ: interferon-γ; IgG: immunoglobulin G; ISEV: international society of extracellular vesicles; lncRNA: long non-coding RNA; LP: lichen planus; LPS: lipopolysaccharides; MHC: major histocompatibility complex; miR: microRNA; MSCs: mesenchymal stem cells; MVB: multivesicular body; MVs: microvesicles; NETs: neutrophil extracellular traps; NK: natural killer; RNA-seq: RNA-sequencing; S. aureus: staphylococcus aureus; SLE: systemic lupus erythematosus; TAMs: tumor-associated macrophages; TCR: T cell receptor; TEM: transmission electron microscope; TGF-β: transforming growth factor-β; Th: T helper; TLR4: Toll-like receptor 4.
Acknowledgements
This work was supported by grants from National Natural Science Foundation of China (81872519, 81673051, and 81703125), and Xijing hospital (XJZT19MJ19).
Competing Interests
The authors have declared that no competing interest exists.
References
1. S ELA, Mager I, Breakefield XO, Wood MJ. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov. 2013;12:347-57
2. Mathieu M, Martin-Jaular L, Lavieu G, Thery C. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat Cell Biol. 2019;21:9-17
3. van Niel G, D'Angelo G, Raposo G. Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol. 2018;19:213-28
4. Fernandez-Messina L, Rodriguez-Galan A, de Yebenes VG, Gutierrez-Vazquez C, Tenreiro S, Seabra MC. et al. Transfer of extracellular vesicle-microRNA controls germinal center reaction and antibody production. EMBO Rep. 2020;21:e48925
5. Keller MD, Ching KL, Liang FX, Dhabaria A, Tam K, Ueberheide BM. et al. Decoy exosomes provide protection against bacterial toxins. Nature. 2020;579:260-4
6. Sjoqvist S, Kasai Y, Shimura D, Ishikawa T, Ali N, Iwata T. et al. Oral keratinocyte-derived exosomes regulate proliferation of fibroblasts and epithelial cells. Biochem Biophys Res Commun. 2019;514:706-12
7. Li Q, Zhao H, Chen W, Huang P, Bi J. Human keratinocyte-derived microvesicle miRNA-21 promotes skin wound healing in diabetic rats through facilitating fibroblast function and angiogenesis. Int J Biochem Cell Biol. 2019;114:105570
8. Angioni R, Liboni C, Herkenne S, Sanchez-Rodriguez R, Borile G, Marcuzzi E. et al. CD73(+) extracellular vesicles inhibit angiogenesis through adenosine A2B receptor signalling. J Extracell Vesicles. 2020;9:1757900
9. Varderidou-Minasian S, Lorenowicz MJ. Mesenchymal stromal/stem cell-derived extracellular vesicles in tissue repair: challenges and opportunities. Theranostics. 2020;10:5979-97
10. Lasser C, Thery C, Buzas EI, Mathivanan S, Zhao W, Gho YS. et al. The International Society for Extracellular Vesicles launches the first massive open online course on extracellular vesicles. J Extracell Vesicles. 2016;5:34299
11. Cheung KL, Jarrett R, Subramaniam S, Salimi M, Gutowska-Owsiak D, Chen YL. et al. Psoriatic T cells recognize neolipid antigens generated by mast cell phospholipase delivered by exosomes and presented by CD1a. J Exp Med. 2016;213:2399-412
12. Jiang M, Fang H, Shao S, Dang E, Zhang J, Qiao P. et al. Keratinocyte exosomes activate neutrophils and enhance skin inflammation in psoriasis. FASEB J. 2019;33:13241-53
13. Kim J, Bin BH, Choi EJ, Lee HG, Lee TR, Cho EG. Staphylococcus aureus-derived extracellular vesicles induce monocyte recruitment by activating human dermal microvascular endothelial cells in vitro. Clin Exp Allergy. 2019;49:68-81
14. Peng Q, Zhang J, Zhou G. Circulating exosomes regulate T-cell-mediated inflammatory response in oral lichen planus. J Oral Pathol Med. 2019;48:143-50
15. Fang H, Shao S, Jiang M, Dang E, Shen S, Zhang J. et al. Proinflammatory role of blister fluid-derived exosomes in bullous pemphigoid. J Pathol. 2018;245:114-25
16. Atehortua L, Rojas M, Vasquez G, Munoz-Vahos CH, Vanegas-Garcia A, Posada-Duque RA. et al. Endothelial activation and injury by microparticles in patients with systemic lupus erythematosus and rheumatoid arthritis. Arthritis Res Ther. 2019;21:34
17. Wang ZY, Yan BX, Zhou Y, Chen XY, Zhang J, Cai SQ. et al. miRNA Profiling of Extracellular Vesicles Reveals Biomarkers for Psoriasis. J Invest Dermatol. 2020 S0022-202X(20)31586-4
18. Lee JY, Park JK, Lee EY, Lee EB, Song YW. Circulating exosomes from patients with systemic lupus erythematosus induce an proinflammatory immune response. Arthritis Res Ther. 2016;18:264
19. Byun JS, Hong SH, Choi JK, Jung JK, Lee HJ. Diagnostic profiling of salivary exosomal microRNAs in oral lichen planus patients. Oral Dis. 2015;21:987-93
20. Merad M, Sathe P, Helft J, Miller J, Mortha A. The dendritic cell lineage: ontogeny and function of dendritic cells and their subsets in the steady state and the inflamed setting. Annu Rev Immunol. 2013;31:563-604
21. Zitvogel L, Regnault A, Lozier A, Wolfers J, Flament C, Tenza D. et al. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat Med. 1998;4:594-600
22. Thery C, Duban L, Segura E, Veron P, Lantz O, Amigorena S. Indirect activation of naive CD4+ T cells by dendritic cell-derived exosomes. Nat Immunol. 2002;3:1156-62
23. Ruhland MK, Roberts EW, Cai E, Mujal AM, Marchuk K, Beppler C. et al. Visualizing Synaptic Transfer of Tumor Antigens among Dendritic Cells. Cancer Cell. 2020;37:786-99 e5
24. Driedonks TAP, van der Grein SG, Ariyurek Y, Buermans HPJ, Jekel H, Chow FWN. et al. Immune stimuli shape the small non-coding transcriptome of extracellular vesicles released by dendritic cells. Cell Mol Life Sci. 2018;75:3857-75
25. Yin W, Ouyang S, Luo Z, Zeng Q, Hu B, Xu L. et al. Immature Exosomes Derived from MicroRNA-146a Overexpressing Dendritic Cells Act as Antigen-Specific Therapy for Myasthenia Gravis. Inflammation. 2017;40:1460-73
26. Segura E, Amigorena S, Thery C. Mature dendritic cells secrete exosomes with strong ability to induce antigen-specific effector immune responses. Blood Cells Mol Dis. 2005;35:89-93
27. Segura E, Nicco C, Lombard B, Veron P, Raposo G, Batteux F. et al. ICAM-1 on exosomes from mature dendritic cells is critical for efficient naive T-cell priming. Blood. 2005;106:216-23
28. Segura E, Guerin C, Hogg N, Amigorena S, Thery C. CD8+ dendritic cells use LFA-1 to capture MHC-peptide complexes from exosomes in vivo. J Immunol. 2007;179:1489-96
29. Liu Q, Rojas-Canales DM, Divito SJ, Shufesky WJ, Stolz DB, Erdos G. et al. Donor dendritic cell-derived exosomes promote allograft-targeting immune response. J Clin Invest. 2016;126:2805-20
30. Ma B, Yang JY, Song WJ, Ding R, Zhang ZC, Ji HC. et al. Combining Exosomes Derived from Immature DCs with Donor Antigen-Specific Treg Cells Induces Tolerance in a Rat Liver Allograft Model. Sci Rep. 2016;6:32971
31. Wahlund CJE, Gucluler G, Hiltbrunner S, Veerman RE, Naslund TI, Gabrielsson S. Exosomes from antigen-pulsed dendritic cells induce stronger antigen-specific immune responses than microvesicles in vivo. Sci Rep. 2017;7:17095
32. Tkach M, Kowal J, Zucchetti AE, Enserink L, Jouve M, Lankar D. et al. Qualitative differences in T-cell activation by dendritic cell-derived extracellular vesicle subtypes. EMBO J. 2017;36:3012-28
33. Zhang Y, Meng J, Zhang L, Ramkrishnan S, Roy S. Extracellular Vesicles with Exosome-like Features Transfer TLRs between Dendritic Cells. Immunohorizons. 2019;3:186-93
34. Choi HW, Suwanpradid J, Kim IH, Staats HF, Haniffa M, MacLeod AS. et al. Perivascular dendritic cells elicit anaphylaxis by relaying allergens to mast cells via microvesicles. Science. 2018;362:eaao0666
35. Feneant L, Levy S, Cocquerel L. CD81 and hepatitis C virus (HCV) infection. Viruses. 2014;6:535-72
36. Martins ST, Kuczera D, Lotvall J, Bordignon J, Alves LR. Characterization of Dendritic Cell-Derived Extracellular Vesicles During Dengue Virus Infection. Front Microbiol. 2018;9:1792
37. Kulkarni R, Prasad A. Exosomes Derived from HIV-1 Infected DCs Mediate Viral trans-Infection via Fibronectin and Galectin-3. Sci Rep. 2017;7:14787
38. Kurosaki T, Shinohara H, Baba Y. B cell signaling and fate decision. Annu Rev Immunol. 2010;28:21-55
39. Raposo G, Nijman HW, Stoorvogel W, Liejendekker R, Harding CV, Melief CJ. et al. B lymphocytes secrete antigen-presenting vesicles. J Exp Med. 1996;183:1161-72
40. Wubbolts R, Leckie RS, Veenhuizen PT, Schwarzmann G, Mobius W, Hoernschemeyer J. et al. Proteomic and biochemical analyses of human B cell-derived exosomes. Potential implications for their function and multivesicular body formation. J Biol Chem. 2003;278:10963-72
41. Papp K, Vegh P, Prechl J, Kerekes K, Kovacs J, Csikos G. et al. B lymphocytes and macrophages release cell membrane deposited C3-fragments on exosomes with T cell response-enhancing capacity. Mol Immunol. 2008;45:2343-51
42. Buschow SI, van Balkom BW, Aalberts M, Heck AJ, Wauben M, Stoorvogel W. MHC class II-associated proteins in B-cell exosomes and potential functional implications for exosome biogenesis. Immunol Cell Biol. 2010;88:851-6
43. Rialland P, Lankar D, Raposo G, Bonnerot C, Hubert P. BCR-bound antigen is targeted to exosomes in human follicular lymphoma B-cells. Biol Cell. 2006;98:491-501
44. Saunderson SC, Schuberth PC, Dunn AC, Miller L, Hock BD, MacKay PA. et al. Induction of exosome release in primary B cells stimulated via CD40 and the IL-4 receptor. J Immunol. 2008;180:8146-52
45. Momen-Heravi F, Bala S, Bukong T, Szabo G. Exosome-mediated delivery of functionally active miRNA-155 inhibitor to macrophages. Nanomedicine. 2014;10:1517-27
46. Klinker MW, Lizzio V, Reed TJ, Fox DA, Lundy SK. Human B Cell-Derived Lymphoblastoid Cell Lines Constitutively Produce Fas Ligand and Secrete MHCII(+)FasL(+) Killer Exosomes. Front Immunol. 2014;5:144
47. Berkani N, Joly P, Golinski ML, Colliou N, Lim A, Larbi A. et al. B-cell depletion induces a shift in self antigen specific B-cell repertoire and cytokine pattern in patients with bullous pemphigoid. Sci Rep. 2019;9:3525
48. Wang S, Wang J, Kumar V, Karnell JL, Naiman B, Gross PS. et al. IL-21 drives expansion and plasma cell differentiation of autoreactive CD11c(hi)T-bet(+) B cells in SLE. Nat Commun. 2018;9:1758
49. Blanchard N, Lankar D, Faure F, Regnault A, Dumont C, Raposo G. et al. TCR activation of human T cells induces the production of exosomes bearing the TCR/CD3/zeta complex. J Immunol. 2002;168:3235-41
50. Alonso R, Mazzeo C, Rodriguez MC, Marsh M, Fraile-Ramos A, Calvo V. et al. Diacylglycerol kinase alpha regulates the formation and polarisation of mature multivesicular bodies involved in the secretion of Fas ligand-containing exosomes in T lymphocytes. Cell Death Differ. 2011;18:1161-73
51. Hedlund M, Nagaeva O, Kargl D, Baranov V, Mincheva-Nilsson L. Thermal- and oxidative stress causes enhanced release of NKG2D ligand-bearing immunosuppressive exosomes in leukemia/lymphoma T and B cells. PLoS One. 2011;6:e16899
52. Xie Y, Zhang H, Li W, Deng Y, Munegowda MA, Chibbar R. et al. Dendritic cells recruit T cell exosomes via exosomal LFA-1 leading to inhibition of CD8+ CTL responses through downregulation of peptide/MHC class I and Fas ligand-mediated cytotoxicity. J Immunol. 2010;185:5268-78
53. Yang J, Dang G, Lu S, Liu H, Ma X, Han L. et al. T-cell-derived extracellular vesicles regulate B-cell IgG production via pyruvate kinase muscle isozyme 2. FASEB J. 2019;33:12780-99
54. Torri A, Carpi D, Bulgheroni E, Crosti MC, Moro M, Gruarin P. et al. Extracellular MicroRNA Signature of Human Helper T Cell Subsets in Health and Autoimmunity. J Biol Chem. 2017;292:2903-15
55. Okoye IS, Coomes SM, Pelly VS, Czieso S, Papayannopoulos V, Tolmachova T. et al. MicroRNA-containing T-regulatory-cell-derived exosomes suppress pathogenic T helper 1 cells. Immunity. 2014;41:89-103
56. Seo N, Shirakura Y, Tahara Y, Momose F, Harada N, Ikeda H. et al. Activated CD8(+) T cell extracellular vesicles prevent tumour progression by targeting of lesional mesenchymal cells. Nat Commun. 2018;9:435
57. Shefler I, Pasmanik-Chor M, Kidron D, Mekori YA, Hershko AY. T cell-derived microvesicles induce mast cell production of IL-24: relevance to inflammatory skin diseases. J Allergy Clin Immunol. 2014;133:217-24 e1-3
58. Lu J, Wu J, Xie F, Tian J, Tang X, Guo H. et al. CD4(+) T Cell-Released Extracellular Vesicles Potentiate the Efficacy of the HBsAg Vaccine by Enhancing B Cell Responses. Adv Sci (Weinh). 2019;6:1802219
59. Garzetti L, Menon R, Finardi A, Bergami A, Sica A, Martino G. et al. Activated macrophages release microvesicles containing polarized M1 or M2 mRNAs. J Leukoc Biol. 2014;95:817-25
60. Milbank E, Soleti R, Martinez E, Lahouel B, Hilairet G, Martinez MC. et al. Microparticles from apoptotic RAW 264.7 macrophage cells carry tumour necrosis factor-alpha functionally active on cardiomyocytes from adult mice. J Extracell Vesicles. 2015;4:28621
61. New SE, Goettsch C, Aikawa M, Marchini JF, Shibasaki M, Yabusaki K. et al. Macrophage-derived matrix vesicles: an alternative novel mechanism for microcalcification in atherosclerotic plaques. Circ Res. 2013;113:72-7
62. Hassani K, Olivier M. Immunomodulatory impact of leishmania-induced macrophage exosomes: a comparative proteomic and functional analysis. PLoS Negl Trop Dis. 2013;7:e2185
63. He C, Hu X, Weston TA, Jung RS, Sandhu J, Huang S. et al. Macrophages release plasma membrane-derived particles rich in accessible cholesterol. Proc Natl Acad Sci U S A. 2018;115:E8499-E508
64. Rajendran RL, Gangadaran P, Seo CH, Kwack MH, Oh JM, Lee HW. et al. Macrophage-Derived Extracellular Vesicle Promotes Hair Growth. Cells. 2020;9:856
65. Ismail N, Wang Y, Dakhlallah D, Moldovan L, Agarwal K, Batte K. et al. Macrophage microvesicles induce macrophage differentiation and miR-223 transfer. Blood. 2013;121:984-95
66. Jiao Y, Li Z, Loughran PA, Fan EK, Scott MJ, Li Y. et al. Frontline Science: Macrophage-derived exosomes promote neutrophil necroptosis following hemorrhagic shock. J Leukoc Biol. 2018;103:175-83
67. Wang C, Zhang C, Liu L, A X, Chen B, Li Y. et al. Macrophage-Derived mir-155-Containing Exosomes Suppress Fibroblast Proliferation and Promote Fibroblast Inflammation during Cardiac Injury. Mol Ther. 2017;25:192-204
68. Wang G, Jin S, Ling X, Li Y, Hu Y, Zhang Y. et al. Proteomic Profiling of LPS-Induced Macrophage-Derived Exosomes Indicates Their Involvement in Acute Liver Injury. Proteomics. 2019;19:e1800274
69. Soni S, Wilson MR, O'Dea KP, Yoshida M, Katbeh U, Woods SJ. et al. Alveolar macrophage-derived microvesicles mediate acute lung injury. Thorax. 2016;71:1020-9
70. Goulielmaki E, Ioannidou A, Tsekrekou M, Stratigi K, Poutakidou IK, Gkirtzimanaki K. et al. Tissue-infiltrating macrophages mediate an exosome-based metabolic reprogramming upon DNA damage. Nat Commun. 2020;11:42
71. Li M, Wang T, Tian H, Wei G, Zhao L, Shi Y. Macrophage-derived exosomes accelerate wound healing through their anti-inflammation effects in a diabetic rat model. Artif Cells Nanomed Biotechnol. 2019;47:3793-803
72. Lee HD, Kim YH, Kim DS. Exosomes derived from human macrophages suppress endothelial cell migration by controlling integrin trafficking. Eur J Immunol. 2014;44:1156-69
73. Uttarkar S, Brembilla NC, Boehncke WH. Regulatory cells in the skin: Pathophysiologic role and potential targets for anti-inflammatory therapies. J Allergy Clin Immunol. 2019;143:1302-10
74. Kolaczkowska E, Kubes P. Neutrophil recruitment and function in health and inflammation. Nat Rev Immunol. 2013;13:159-75
75. Stein JM, Luzio JP. Ectocytosis caused by sublytic autologous complement attack on human neutrophils. The sorting of endogenous plasma-membrane proteins and lipids into shed vesicles. Biochem J. 1991;274:381-6
76. Timar CI, Lorincz AM, Csepanyi-Komi R, Valyi-Nagy A, Nagy G, Buzas EI. et al. Antibacterial effect of microvesicles released from human neutrophilic granulocytes. Blood. 2013;121:510-8
77. Gasser O, Hess C, Miot S, Deon C, Sanchez JC, Schifferli JA. Characterisation and properties of ectosomes released by human polymorphonuclear neutrophils. Exp Cell Res. 2003;285:243-57
78. Gasser O, Schifferli JA. Microparticles released by human neutrophils adhere to erythrocytes in the presence of complement. Exp Cell Res. 2005;307:381-7
79. Pluskota E, Woody NM, Szpak D, Ballantyne CM, Soloviev DA, Simon DI. et al. Expression, activation, and function of integrin alphaMbeta2 (Mac-1) on neutrophil-derived microparticles. Blood. 2008;112:2327-35
80. Slater TW, Finkielsztein A, Mascarenhas LA, Mehl LC, Butin-Israeli V, Sumagin R. Neutrophil Microparticles Deliver Active Myeloperoxidase to Injured Mucosa To Inhibit Epithelial Wound Healing. J Immunol. 2017;198:2886-97
81. Dalli J, Montero-Melendez T, Norling LV, Yin X, Hinds C, Haskard D. et al. Heterogeneity in neutrophil microparticles reveals distinct proteome and functional properties. Mol Cell Proteomics. 2013;12:2205-19
82. Shao S, Fang H, Zhang J, Jiang M, Xue K, Ma J. et al. Neutrophil exosomes enhance the skin autoinflammation in generalized pustular psoriasis via activating keratinocytes. FASEB J. 2019;33:6813-28
83. Lim K, Hyun YM, Lambert-Emo K, Capece T, Bae S, Miller R. et al. Neutrophil trails guide influenza-specific CD8(+) T cells in the airways. Science. 2015;349:aaa4352
84. Vargas A, Roux-Dalvai F, Droit A, Lavoie JP. Neutrophil-Derived Exosomes: A New Mechanism Contributing to Airway Smooth Muscle Remodeling. Am J Respir Cell Mol Biol. 2016;55:450-61
85. Li L, Zuo X, Liu D, Luo H, Zhu H. The profiles of miRNAs and lncRNAs in peripheral blood neutrophils exosomes of diffuse cutaneous systemic sclerosis. J Dermatol Sci. 2020;98:88-97
86. Shopova IA, Belyaev I, Dasari P, Jahreis S, Stroe MC, Cseresnyes Z. et al. Human Neutrophils Produce Antifungal Extracellular Vesicles against Aspergillus fumigatus. mBio. 2020;11:e00596-20
87. Rossaint J, Kuhne K, Skupski J, Van Aken H, Looney MR, Hidalgo A. et al. Directed transport of neutrophil-derived extracellular vesicles enables platelet-mediated innate immune response. Nat Commun. 2016;7:13464
88. Alvarez-Jimenez VD, Leyva-Paredes K, Garcia-Martinez M, Vazquez-Flores L, Garcia-Paredes VG, Campillo-Navarro M. et al. Extracellular Vesicles Released from Mycobacterium tuberculosis-Infected Neutrophils Promote Macrophage Autophagy and Decrease Intracellular Mycobacterial Survival. Front Immunol. 2018;9:272
89. Mesri M, Altieri DC. Leukocyte microparticles stimulate endothelial cell cytokine release and tissue factor induction in a JNK1 signaling pathway. J Biol Chem. 1999;274:23111-8
90. Hong Y, Eleftheriou D, Hussain AA, Price-Kuehne FE, Savage CO, Jayne D. et al. Anti-neutrophil cytoplasmic antibodies stimulate release of neutrophil microparticles. J Am Soc Nephrol. 2012;23:49-62
91. Genschmer KR, Russell DW, Lal C, Szul T, Bratcher PE, Noerager BD. et al. Activated PMN Exosomes: Pathogenic Entities Causing Matrix Destruction and Disease in the Lung. Cell. 2019;176:113-26 e15
92. Headland SE, Jones HR, Norling LV, Kim A, Souza PR, Corsiero E. et al. Neutrophil-derived microvesicles enter cartilage and protect the joint in inflammatory arthritis. Sci Transl Med. 2015;7:315ra190
93. Pliyev BK, Kalintseva MV, Abdulaeva SV, Yarygin KN, Savchenko VG. Neutrophil microparticles modulate cytokine production by natural killer cells. Cytokine. 2014;65:126-9
94. Eken C, Gasser O, Zenhaeusern G, Oehri I, Hess C, Schifferli JA. Polymorphonuclear neutrophil-derived ectosomes interfere with the maturation of monocyte-derived dendritic cells. J Immunol. 2008;180:817-24
95. Eken C, Sadallah S, Martin PJ, Treves S, Schifferli JA. Ectosomes of polymorphonuclear neutrophils activate multiple signaling pathways in macrophages. Immunobiology. 2013;218:382-92
96. Gasser O, Schifferli JA. Activated polymorphonuclear neutrophils disseminate anti-inflammatory microparticles by ectocytosis. Blood. 2004;104:2543-8
97. Rhys HI, Dell'Accio F, Pitzalis C, Moore A, Norling LV, Perretti M. Neutrophil Microvesicles from Healthy Control and Rheumatoid Arthritis Patients Prevent the Inflammatory Activation of Macrophages. EBioMedicine. 2018;29:60-9
98. Cumpelik A, Ankli B, Zecher D, Schifferli JA. Neutrophil microvesicles resolve gout by inhibiting C5a-mediated priming of the inflammasome. Ann Rheum Dis. 2016;75:1236-45
99. Pitanga TN, de Aragao Franca L, Rocha VC, Meirelles T, Borges VM, Goncalves MS. et al. Neutrophil-derived microparticles induce myeloperoxidase-mediated damage of vascular endothelial cells. BMC Cell Biol. 2014;15:21
100. Wang L, Luo H, Chen X, Jiang Y, Huang Q. Functional characterization of S100A8 and S100A9 in altering monolayer permeability of human umbilical endothelial cells. PLoS One. 2014;9:e90472
101. Manchanda K, Kolarova H, Kerkenpass C, Mollenhauer M, Vitecek J, Rudolph V. et al. MPO (Myeloperoxidase) Reduces Endothelial Glycocalyx Thickness Dependent on Its Cationic Charge. Arterioscler Thromb Vasc Biol. 2018;38:1859-67
102. Cohen-Mazor M, Mazor R, Kristal B, Sela S. Elastase and cathepsin G from primed leukocytes cleave vascular endothelial cadherin in hemodialysis patients. Biomed Res Int. 2014;2014:459640
103. Cristante E, McArthur S, Mauro C, Maggioli E, Romero IA, Wylezinska-Arridge M. et al. Identification of an essential endogenous regulator of blood-brain barrier integrity, and its pathological and therapeutic implications. Proc Natl Acad Sci U S A. 2013;110:832-41
104. Dalli J, Norling LV, Renshaw D, Cooper D, Leung KY, Perretti M. Annexin 1 mediates the rapid anti-inflammatory effects of neutrophil-derived microparticles. Blood. 2008;112:2512-9
105. Kouroupis D, Sanjurjo-Rodriguez C, Jones E, Correa D. Mesenchymal Stem Cell Functionalization for Enhanced Therapeutic Applications. Tissue Eng Part B Rev. 2019;25:55-77
106. Fang S, Xu C, Zhang Y, Xue C, Yang C, Bi H. et al. Umbilical Cord-Derived Mesenchymal Stem Cell-Derived Exosomal MicroRNAs Suppress Myofibroblast Differentiation by Inhibiting the Transforming Growth Factor-beta/SMAD2 Pathway During Wound Healing. Stem Cells Transl Med. 2016;5:1425-39
107. Baglio SR, Rooijers K, Koppers-Lalic D, Verweij FJ, Perez Lanzon M, Zini N. et al. Human bone marrow- and adipose-mesenchymal stem cells secrete exosomes enriched in distinctive miRNA and tRNA species. Stem Cell Res Ther. 2015;6:127
108. Eirin A, Riester SM, Zhu XY, Tang H, Evans JM, O'Brien D. et al. MicroRNA and mRNA cargo of extracellular vesicles from porcine adipose tissue-derived mesenchymal stem cells. Gene. 2014;551:55-64
109. Kim HS, Choi DY, Yun SJ, Choi SM, Kang JW, Jung JW. et al. Proteomic analysis of microvesicles derived from human mesenchymal stem cells. J Proteome Res. 2012;11:839-49
110. Anderson JD, Johansson HJ, Graham CS, Vesterlund M, Pham MT, Bramlett CS. et al. Comprehensive Proteomic Analysis of Mesenchymal Stem Cell Exosomes Reveals Modulation of Angiogenesis via Nuclear Factor-KappaB Signaling. Stem Cells. 2016;34:601-13
111. van Balkom BWM, Gremmels H, Giebel B, Lim SK. Proteomic Signature of Mesenchymal Stromal Cell-Derived Small Extracellular Vesicles. Proteomics. 2019;19:e1800163
112. La Greca A, Solari C, Furmento V, Lombardi A, Biani MC, Aban C. et al. Extracellular vesicles from pluripotent stem cell-derived mesenchymal stem cells acquire a stromal modulatory proteomic pattern during differentiation. Exp Mol Med. 2018;50:119
113. Bruno S, Grange C, Deregibus MC, Calogero RA, Saviozzi S, Collino F. et al. Mesenchymal stem cell-derived microvesicles protect against acute tubular injury. J Am Soc Nephrol. 2009;20:1053-67
114. Bruno S, Tapparo M, Collino F, Chiabotto G, Deregibus MC, Soares Lindoso R. et al. Renal Regenerative Potential of Different Extracellular Vesicle Populations Derived from Bone Marrow Mesenchymal Stromal Cells. Tissue Eng Part A. 2017;23:1262-73
115. Choi HY, Moon SJ, Ratliff BB, Ahn SH, Jung A, Lee M. et al. Microparticles from kidney-derived mesenchymal stem cells act as carriers of proangiogenic signals and contribute to recovery from acute kidney injury. PLoS One. 2014;9:e87853
116. Zou X, Zhang G, Cheng Z, Yin D, Du T, Ju G. et al. Microvesicles derived from human Wharton's Jelly mesenchymal stromal cells ameliorate renal ischemia-reperfusion injury in rats by suppressing CX3CL1. Stem Cell Res Ther. 2014;5:40
117. Cao H, Cheng Y, Gao H, Zhuang J, Zhang W, Bian Q. et al. In vivo Tracking of Mesenchymal Stem Cell-Derived Extracellular Vesicles Improving Mitochondrial Function in Renal Ischemia-Reperfusion Injury. ACS Nano. 2020;14:4014-26
118. Li T, Yan Y, Wang B, Qian H, Zhang X, Shen L. et al. Exosomes derived from human umbilical cord mesenchymal stem cells alleviate liver fibrosis. Stem Cells Dev. 2013;22:845-54
119. Tan CY, Lai RC, Wong W, Dan YY, Lim SK, Ho HK. Mesenchymal stem cell-derived exosomes promote hepatic regeneration in drug-induced liver injury models. Stem Cell Res Ther. 2014;5:76
120. Lou G, Yang Y, Liu F, Ye B, Chen Z, Zheng M. et al. MiR-122 modification enhances the therapeutic efficacy of adipose tissue-derived mesenchymal stem cells against liver fibrosis. J Cell Mol Med. 2017;21:2963-73
121. Tamura R, Uemoto S, Tabata Y. Immunosuppressive effect of mesenchymal stem cell-derived exosomes on a concanavalin A-induced liver injury model. Inflamm Regen. 2016;36:26
122. Yan B, Zhang Y, Liang C, Liu B, Ding F, Wang Y. et al. Stem cell-derived exosomes prevent pyroptosis and repair ischemic muscle injury through a novel exosome/circHIPK3/ FOXO3a pathway. Theranostics. 2020;10:6728-42
123. Cho BS, Kim JO, Ha DH, Yi YW. Exosomes derived from human adipose tissue-derived mesenchymal stem cells alleviate atopic dermatitis. Stem Cell Res Ther. 2018;9:187
124. Nestle FO, Di Meglio P, Qin JZ, Nickoloff BJ. Skin immune sentinels in health and disease. Nat Rev Immunol. 2009;9:679-91
125. Mangino G, Iuliano M, Carlomagno S, Bernardini N, Rosa P, Chiantore MV. et al. Interleukin-17A affects extracellular vesicles release and cargo in human keratinocytes. Exp Dermatol. 2019;28:1066-73
126. Cai XW, Zhu R, Ran L, Li YQ, Huang K, Peng J. et al. A novel noncontact communication between human keratinocytes and T cells: Exosomes derived from keratinocytes support superantigeninduced proliferation of resting T cells. Mol Med Rep. 2017;16:7032-8
127. Than UTT, Guanzon D, Broadbent JA, Leavesley DI, Salomon C, Parker TJ. Differential Expression of Keratinocyte-Derived Extracellular Vesicle Mirnas Discriminate Exosomes From Apoptotic Bodies and Microvesicles. Front Endocrinol (Lausanne). 2018;9:535
128. Buth H, Wolters B, Hartwig B, Meier-Bornheim R, Veith H, Hansen M. et al. HaCaT keratinocytes secrete lysosomal cysteine proteinases during migration. Eur J Cell Biol. 2004;83:781-95
129. Chavez-Munoz C, Kilani RT, Ghahary A. Profile of exosomes related proteins released by differentiated and undifferentiated human keratinocytes. J Cell Physiol. 2009;221:221-31
130. Lo Cicero A, Delevoye C, Gilles-Marsens F, Loew D, Dingli F, Guere C. et al. Exosomes released by keratinocytes modulate melanocyte pigmentation. Nat Commun. 2015;6:7506
131. Kotzerke K, Mempel M, Aung T, Wulf GG, Urlaub H, Wenzel D. et al. Immunostimulatory activity of murine keratinocyte-derived exosomes. Exp Dermatol. 2013;22:650-5
132. Lynch MD, Watt FM. Fibroblast heterogeneity: implications for human disease. J Clin Invest. 2018;128:26-35
133. Bang C, Batkai S, Dangwal S, Gupta SK, Foinquinos A, Holzmann A. et al. Cardiac fibroblast-derived microRNA passenger strand-enriched exosomes mediate cardiomyocyte hypertrophy. J Clin Invest. 2014;124:2136-46
134. Haj-Salem I, Plante S, Gounni AS, Rouabhia M, Chakir J. Fibroblast-derived exosomes promote epithelial cell proliferation through TGF-beta2 signalling pathway in severe asthma. Allergy. 2018;73:178-86
135. Nakamura K, Jinnin M, Harada M, Kudo H, Nakayama W, Inoue K. et al. Altered expression of CD63 and exosomes in scleroderma dermal fibroblasts. J Dermatol Sci. 2016;84:30-9
136. Terlecki-Zaniewicz L, Pils V, Bobbili MR, Lammermann I, Perrotta I, Grillenberger T. et al. Extracellular Vesicles in Human Skin: Cross-Talk from Senescent Fibroblasts to Keratinocytes by miRNAs. J Invest Dermatol. 2019;139:2425-36 e5
137. Deng M, Yu Z, Li D, Wang X, Zhou G, Liu W. et al. Human umbilical cord mesenchymal stem cell-derived and dermal fibroblast-derived extracellular vesicles protect dermal fibroblasts from ultraviolet radiation-induced photoaging in vitro. Photochem Photobiol Sci. 2020;19:406-14
138. de Candia P, De Rosa V, Gigantino V, Botti G, Ceriello A, Matarese G. Immunometabolism of human autoimmune diseases: from metabolites to extracellular vesicles. FEBS Lett. 2017;591:3119-34
139. Connolly KD, Guschina IA, Yeung V, Clayton A, Draman MS, Von Ruhland C. et al. Characterisation of adipocyte-derived extracellular vesicles released pre- and post-adipogenesis. J Extracell Vesicles. 2015;4:29159
140. Flaherty SE 3rd, Grijalva A, Xu X, Ables E, Nomani A, Ferrante AW Jr. A lipase-independent pathway of lipid release and immune modulation by adipocytes. Science. 2019;363:989-93
141. Zhang X, Chen L, Xiao B, Liu H, Su Y. Circ_0075932 in adipocyte-derived exosomes induces inflammation and apoptosis in human dermal keratinocytes by directly binding with PUM2 and promoting PUM2-mediated activation of AuroraA/NF-kappaB pathway. Biochem Biophys Res Commun. 2019;511:551-8
142. Mutt SJ, Herzig KH. Adipocyte-derived extracellular vesicles as new communication signals in the regulation of food intake. Acta Physiol (Oxf). 2020;228:e13411
143. Kranendonk ME, Visseren FL, van Herwaarden JA, Nolte-'t Hoen EN, de Jager W, Wauben MH. et al. Effect of extracellular vesicles of human adipose tissue on insulin signaling in liver and muscle cells. Obesity (Silver Spring). 2014;22:2216-23
144. Hotta K, Funahashi T, Arita Y, Takahashi M, Matsuda M, Okamoto Y. et al. Plasma concentrations of a novel, adipose-specific protein, adiponectin, in type 2 diabetic patients. Arterioscler Thromb Vasc Biol. 2000;20:1595-9
145. Wilk S, Scheibenbogen C, Bauer S, Jenke A, Rother M, Guerreiro M. et al. Adiponectin is a negative regulator of antigen-activated T cells. Eur J Immunol. 2011;41:2323-32
146. Durcin M, Fleury A, Taillebois E, Hilairet G, Krupova Z, Henry C. et al. Characterisation of adipocyte-derived extracellular vesicle subtypes identifies distinct protein and lipid signatures for large and small extracellular vesicles. J Extracell Vesicles. 2017;6:1305677
147. Jin SE, Sung JH. Hair regeneration using adipose-derived stem cells. Histol Histopathol. 2016;31:249-56
148. Li CJ, Fang QH, Liu ML, Lin JN. Current understanding of the role of Adipose-derived Extracellular Vesicles in Metabolic Homeostasis and Diseases: Communication from the distance between cells/tissues. Theranostics. 2020;10:7422-35
149. Eguchi A, Lazic M, Armando AM, Phillips SA, Katebian R, Maraka S. et al. Circulating adipocyte-derived extracellular vesicles are novel markers of metabolic stress. J Mol Med (Berl). 2016;94:1241-53
150. Gao J, Li X, Wang Y, Cao Y, Yao D, Sun L. et al. Adipocyte-derived extracellular vesicles modulate appetite and weight through mTOR signalling in the hypothalamus. Acta Physiol (Oxf). 2020;228:e13339
151. Yu Y, Du H, Wei S, Feng L, Li J, Yao F. et al. Adipocyte-Derived Exosomal MiR-27a Induces Insulin Resistance in Skeletal Muscle Through Repression of PPARgamma. Theranostics. 2018;8:2171-88
152. Wei M, Gao X, Liu L, Li Z, Wan Z, Dong Y. et al. Visceral Adipose Tissue Derived Exosomes Exacerbate Colitis Severity via Pro-inflammatory MiRNAs in High Fat Diet Fed Mice. ACS Nano. 2020;14:5099-110
153. Gan L, Xie D, Liu J, Bond Lau W, Christopher TA, Lopez B. et al. Small Extracellular Microvesicles Mediated Pathological Communications Between Dysfunctional Adipocytes and Cardiomyocytes as a Novel Mechanism Exacerbating Ischemia/Reperfusion Injury in Diabetic Mice. Circulation. 2020;141:968-83
154. Li X, Ballantyne LL, Yu Y, Funk CD. Perivascular adipose tissue-derived extracellular vesicle miR-221-3p mediates vascular remodeling. FASEB J. 2019;33:12704-22
155. Wadey RM, Connolly KD, Mathew D, Walters G, Rees DA, James PE. Inflammatory adipocyte-derived extracellular vesicles promote leukocyte attachment to vascular endothelial cells. Atherosclerosis. 2019;283:19-27
156. Perera GK, Di Meglio P, Nestle FO. Psoriasis. Annu Rev Pathol. 2012;7:385-422
157. Tamagawa-Mineoka R, Katoh N, Kishimoto S. Platelet activation in patients with psoriasis: increased plasma levels of platelet-derived microparticles and soluble P-selectin. J Am Acad Dermatol. 2010;62:621-6
158. Pelletier F, Garnache-Ottou F, Angelot F, Biichle S, Vidal C, Humbert P. et al. Increased levels of circulating endothelial-derived microparticles and small-size platelet-derived microparticles in psoriasis. J Invest Dermatol. 2011;131:1573-6
159. Martinez-Sales V, Vila V, Ricart JM, Vaya A, Todoli J, Nnnez C. et al. Increased circulating endothelial cells and microparticles in patients with psoriasis. Clin Hemorheol Microcirc. 2015;60:283-90
160. Takeshita J, Mohler ER, Krishnamoorthy P, Moore J, Rogers WT, Zhang L. et al. Endothelial cell-, platelet-, and monocyte/macrophage-derived microparticles are elevated in psoriasis beyond cardiometabolic risk factors. J Am Heart Assoc. 2014;3:e000507
161. Jacquin-Porretaz C, Cordonnier M, Nardin C, Boullerot L, Chanteloup G, Vautrot V. et al. Increased Levels of Interleukin-17A Exosomes in Psoriasis. Acta Derm Venereol. 2019;99:1143-7
162. Ho JC, Lee CH, Lin SH. No Significant Reduction of Circulating Endothelial-Derived and Platelet-Derived Microparticles in Patients with Psoriasis Successfully Treated with Anti-IL12/23. Biomed Res Int. 2016;2016:3242143
163. Pasquali L, Svedbom A, Srivastava A, Rosen E, Lindqvist U, Stahle M. et al. Circulating microRNAs in extracellular vesicles as potential biomarkers for psoriatic arthritis in patients with psoriasis. J Eur Acad Dermatol Venereol. 2020;34:1248-1256
164. Jiang M, Fang H, Dang E, Zhang J, Qiao P, Yu C. et al. Small extracellular vesicles containing miR-381-3p from keratinocytes promotes Th1/Th17 polarization in psoriasis. J Invest Dermatol. 2020 S0022-202X(20)31938-2
165. Marton N, Kovacs OT, Baricza E, Kittel A, Gyori D, Mocsai A. et al. Extracellular vesicles regulate the human osteoclastogenesis: divergent roles in discrete inflammatory arthropathies. Cell Mol Life Sci. 2017;74:3599-611
166. Weidinger S, Novak N. Atopic dermatitis. Lancet. 2016;387:1109-22
167. Dainichi T, Kitoh A, Otsuka A, Nakajima S, Nomura T, Kaplan DH. et al. The epithelial immune microenvironment (EIME) in atopic dermatitis and psoriasis. Nat Immunol. 2018;19:1286-98
168. Hong SW, Kim MR, Lee EY, Kim JH, Kim YS, Jeon SG. et al. Extracellular vesicles derived from Staphylococcus aureus induce atopic dermatitis-like skin inflammation. Allergy. 2011;66:351-9
169. Jun SH, Lee JH, Kim SI, Choi CW, Park TI, Jung HR. et al. Staphylococcus aureus-derived membrane vesicles exacerbate skin inflammation in atopic dermatitis. Clin Exp Allergy. 2017;47:85-96
170. Hong SW, Choi EB, Min TK, Kim JH, Kim MH, Jeon SG. et al. An important role of alpha-hemolysin in extracellular vesicles on the development of atopic dermatitis induced by Staphylococcus aureus. PLoS One. 2014;9:e100499
171. Kwon HI, Jeong NH, Jun SH, Son JH, Kim S, Jeon H. et al. Thymol attenuates the worsening of atopic dermatitis induced by Staphylococcus aureus membrane vesicles. Int Immunopharmacol. 2018;59:301-9
172. Kim MH, Choi SJ, Choi HI, Choi JP, Park HK, Kim EK. et al. Lactobacillus plantarum-derived Extracellular Vesicles Protect Atopic Dermatitis Induced by Staphylococcus aureus-derived Extracellular Vesicles. Allergy Asthma Immunol Res. 2018;10:516-32
173. Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826
174. Shao S, Tsoi LC, Sarkar MK, Xing X, Xue K, Uppala R. et al. IFN-gamma enhances cell-mediated cytotoxicity against keratinocytes via JAK2/STAT1 in lichen planus. Sci Transl Med. 2019;11:eaav7561
175. Schmidt E, Zillikens D. Pemphigoid diseases. Lancet. 2013;381:320-32
176. Verraes S, Hornebeck W, Polette M, Borradori L, Bernard P. Respective contribution of neutrophil elastase and matrix metalloproteinase 9 in the degradation of BP180 (type XVII collagen) in human bullous pemphigoid. J Invest Dermatol. 2001;117:1091-6
177. Riani M, Le Jan S, Plee J, Durlach A, Le Naour R, Haegeman G. et al. Bullous pemphigoid outcome is associated with CXCL10-induced matrix metalloproteinase 9 secretion from monocytes and neutrophils but not lymphocytes. J Allergy Clin Immunol. 2017;139:863-72 e3
178. Tsokos GC. Systemic lupus erythematosus. N Engl J Med. 2011;365:2110-21
179. Mobarrez F, Vikerfors A, Gustafsson JT, Gunnarsson I, Zickert A, Larsson A. et al. Microparticles in the blood of patients with systemic lupus erythematosus (SLE): phenotypic characterization and clinical associations. Sci Rep. 2016;6:36025
180. Ullal AJ, Reich CF 3rd, Clowse M, Criscione-Schreiber LG, Tochacek M, Monestier M. et al. Microparticles as antigenic targets of antibodies to DNA and nucleosomes in systemic lupus erythematosus. J Autoimmun. 2011;36:173-80
181. Nielsen CT, Ostergaard O, Johnsen C, Jacobsen S, Heegaard NH. Distinct features of circulating microparticles and their relationship to clinical manifestations in systemic lupus erythematosus. Arthritis Rheum. 2011;63:3067-77
182. Nielsen CT, Ostergaard O, Stener L, Iversen LV, Truedsson L, Gullstrand B. et al. Increased IgG on cell-derived plasma microparticles in systemic lupus erythematosus is associated with autoantibodies and complement activation. Arthritis Rheum. 2012;64:1227-36
183. Fortin PR, Cloutier N, Bissonnette V, Aghdassi E, Eder L, Simonyan D. et al. Distinct Subtypes of Microparticle-containing Immune Complexes Are Associated with Disease Activity, Damage, and Carotid Intima-media Thickness in Systemic Lupus Erythematosus. J Rheumatol. 2016;43:2019-25
184. Ostergaard O, Nielsen CT, Iversen LV, Tanassi JT, Knudsen S, Jacobsen S. et al. Unique protein signature of circulating microparticles in systemic lupus erythematosus. Arthritis Rheum. 2013;65:2680-90
185. Mobarrez F, Fuzzi E, Gunnarsson I, Larsson A, Eketjall S, Pisetsky DS. et al. Microparticles in the blood of patients with SLE: Size, content of mitochondria and role in circulating immune complexes. J Autoimmun. 2019;102:142-9
186. Perez-Hernandez J, Forner MJ, Pinto C, Chaves FJ, Cortes R, Redon J. Increased Urinary Exosomal MicroRNAs in Patients with Systemic Lupus Erythematosus. PLoS One. 2015;10:e0138618
187. Winberg LK, Jacobsen S, Nielsen CH. Microparticles from patients with systemic lupus erythematosus induce production of reactive oxygen species and degranulation of polymorphonuclear leukocytes. Arthritis Res Ther. 2017;19:230
188. Salvi V, Gianello V, Busatto S, Bergese P, Andreoli L, D'Oro U. et al. Exosome-delivered microRNAs promote IFN-alpha secretion by human plasmacytoid DCs via TLR7. JCI Insight. 2018;3:e98204
189. Dong C, Zhou Q, Fu T, Zhao R, Yang J, Kong X. et al. Circulating Exosomes Derived-miR-146a from Systemic Lupus Erythematosus Patients Regulates Senescence of Mesenchymal Stem Cells. Biomed Res Int. 2019;2019:6071308
190. Burbano C, Villar-Vesga J, Orejuela J, Munoz C, Vanegas A, Vasquez G. et al. Potential Involvement of Platelet-Derived Microparticles and Microparticles Forming Immune Complexes during Monocyte Activation in Patients with Systemic Lupus Erythematosus. Front Immunol. 2018;9:322
191. Rodrigues M, Kosaric N, Bonham CA, Gurtner GC. Wound Healing: A Cellular Perspective. Physiol Rev. 2019;99:665-706
192. Martin P, Nunan R. Cellular and molecular mechanisms of repair in acute and chronic wound healing. Br J Dermatol. 2015;173:370-8
193. Powers JG, Higham C, Broussard K, Phillips TJ. Wound healing and treating wounds: Chronic wound care and management. J Am Acad Dermatol. 2016;74:607-25 quiz 25-6
194. Wilgus TA, Roy S, McDaniel JC. Neutrophils and Wound Repair: Positive Actions and Negative Reactions. Adv Wound Care (New Rochelle). 2013;2:379-88
195. He L, Marneros AG. Macrophages are essential for the early wound healing response and the formation of a fibrovascular scar. Am J Pathol. 2013;182:2407-17
196. Weller K, Foitzik K, Paus R, Syska W, Maurer M. Mast cells are required for normal healing of skin wounds in mice. FASEB J. 2006;20:2366-8
197. Gregorio J, Meller S, Conrad C, Di Nardo A, Homey B, Lauerma A. et al. Plasmacytoid dendritic cells sense skin injury and promote wound healing through type I interferons. J Exp Med. 2010;207:2921-30
198. Jameson J, Ugarte K, Chen N, Yachi P, Fuchs E, Boismenu R. et al. A role for skin gammadelta T cells in wound repair. Science. 2002;296:747-9
199. Plikus MV, Guerrero-Juarez CF, Ito M, Li YR, Dedhia PH, Zheng Y. et al. Regeneration of fat cells from myofibroblasts during wound healing. Science. 2017;355:748-52
200. McKay TB, Hutcheon AEK, Zieske JD, Ciolino JB. Extracellular Vesicles Secreted by Corneal Epithelial Cells Promote Myofibroblast Differentiation. Cells. 2020;9:1080
201. Berckmans RJ, Sturk A, van Tienen LM, Schaap MC, Nieuwland R. Cell-derived vesicles exposing coagulant tissue factor in saliva. Blood. 2011;117:3172-80
202. Yin H, Chen CY, Liu YW, Tan YJ, Deng ZL, Yang F. et al. Synechococcus elongatus PCC7942 secretes extracellular vesicles to accelerate cutaneous wound healing by promoting angiogenesis. Theranostics. 2019;9:2678-93
203. Shabbir A, Cox A, Rodriguez-Menocal L, Salgado M, Van Badiavas E. Mesenchymal Stem Cell Exosomes Induce Proliferation and Migration of Normal and Chronic Wound Fibroblasts, and Enhance Angiogenesis In vitro. Stem Cells Dev. 2015;24:1635-47
204. Guo SC, Tao SC, Yin WJ, Qi X, Yuan T, Zhang CQ. Exosomes derived from platelet-rich plasma promote the re-epithelization of chronic cutaneous wounds via activation of YAP in a diabetic rat model. Theranostics. 2017;7:81-96
205. Sinha M, Sen CK, Singh K, Das A, Ghatak S, Rhea B. et al. Direct conversion of injury-site myeloid cells to fibroblast-like cells of granulation tissue. Nat Commun. 2018;9:936
206. Zeng T, Wang X, Wang W, Feng Q, Lao G, Liang Y. et al. Endothelial cell-derived small extracellular vesicles suppress cutaneous wound healing through regulating fibroblasts autophagy. Clin Sci (Lond). 2019;133:CS20190008
207. Leszczynska A, Kulkarni M, Ljubimov AV, Saghizadeh M. Exosomes from normal and diabetic human corneolimbal keratocytes differentially regulate migration, proliferation and marker expression of limbal epithelial cells. Sci Rep. 2018;8:15173
208. Parker B, Al-Husain A, Pemberton P, Yates AP, Ho P, Gorodkin R. et al. Suppression of inflammation reduces endothelial microparticles in active systemic lupus erythematosus. Ann Rheum Dis. 2014;73:1144-50
209. Tangtanatakul P, Klinchanhom S, Sodsai P, Sutichet T, Promjeen C, Avihingsanon Y. et al. Down-regulation of let-7a and miR-21 in urine exosomes from lupus nephritis patients during disease flare. Asian Pac J Allergy Immunol. 2019;37:189-97
210. Yang J, McDowell A, Seo H, Kim S, Min TK, Jee YK. et al. Diagnostic Models for Atopic Dermatitis Based on Serum Microbial Extracellular Vesicle Metagenomic Analysis: A Pilot Study. Allergy Asthma Immunol Res. 2020;12:792-805
211. Liu S, Liu D, Chen C, Hamamura K, Moshaverinia A, Yang R. et al. MSC Transplantation Improves Osteopenia via Epigenetic Regulation of Notch Signaling in Lupus. Cell Metab. 2015;22:606-18
212. Kordelas L, Rebmann V, Ludwig AK, Radtke S, Ruesing J, Doeppner TR. et al. MSC-derived exosomes: a novel tool to treat therapy-refractory graft-versus-host disease. Leukemia. 2014;28:970-3
213. Hu Y, Rao SS, Wang ZX, Cao J, Tan YJ, Luo J. et al. Exosomes from human umbilical cord blood accelerate cutaneous wound healing through miR-21-3p-mediated promotion of angiogenesis and fibroblast function. Theranostics. 2018;8:169-84
214. Sjoqvist S, Ishikawa T, Shimura D, Kasai Y, Imafuku A, Bou-Ghannam S. et al. Exosomes derived from clinical-grade oral mucosal epithelial cell sheets promote wound healing. J Extracell Vesicles. 2019;8:1565264
215. Chen CY, Rao SS, Ren L, Hu XK, Tan YJ, Hu Y. et al. Exosomal DMBT1 from human urine-derived stem cells facilitates diabetic wound repair by promoting angiogenesis. Theranostics. 2018;8:1607-23
216. Samaeekia R, Rabiee B, Putra I, Shen X, Park YJ, Hematti P. et al. Effect of Human Corneal Mesenchymal Stromal Cell-derived Exosomes on Corneal Epithelial Wound Healing. Invest Ophthalmol Vis Sci. 2018;59:5194-200
217. Kou X, Xu X, Chen C, Sanmillan ML, Cai T, Zhou Y. et al. The Fas/Fap-1/Cav-1 complex regulates IL-1RA secretion in mesenchymal stem cells to accelerate wound healing. Sci Transl Med. 2018;10:eaai8524
218. Rosca AM, Tutuianu R, Titorencu ID. Mesenchymal stromal cells derived exosomes as tools for chronic wound healing therapy. Rom J Morphol Embryol. 2018;59:655-62
219. Wang C, Wang M, Xu T, Zhang X, Lin C, Gao W. et al. Engineering Bioactive Self-Healing Antibacterial Exosomes Hydrogel for Promoting Chronic Diabetic Wound Healing and Complete Skin Regeneration. Theranostics. 2019;9:65-76
220. Henriques-Antunes H, Cardoso RMS, Zonari A, Correia J, Leal EC, Jimenez-Balsa A. et al. The Kinetics of Small Extracellular Vesicle Delivery Impacts Skin Tissue Regeneration. ACS Nano. 2019;13:8694-707
221. Silva AKA, Perretta S, Perrod G, Pidial L, Lindner V, Carn F. et al. Thermoresponsive Gel Embedded with Adipose Stem-Cell-Derived Extracellular Vesicles Promotes Esophageal Fistula Healing in a Thermo-Actuated Delivery Strategy. ACS Nano. 2018;12:9800-14
222. Escudier B, Dorval T, Chaput N, Andre F, Caby MP, Novault S. et al. Vaccination of metastatic melanoma patients with autologous dendritic cell (DC) derived-exosomes: results of thefirst phase I clinical trial. J Transl Med. 2005;3:10
223. Morse MA, Garst J, Osada T, Khan S, Hobeika A, Clay TM. et al. A phase I study of dexosome immunotherapy in patients with advanced non-small cell lung cancer. J Transl Med. 2005;3:9
224. Elsharkasy OM, Nordin JZ, Hagey DW, de Jong OG, Schiffelers RM, Andaloussi SE. et al. Extracellular vesicles as drug delivery systems: Why and how? Adv Drug Deliv Rev. 2020 S0169-409X(20)30024-7
225. Kim MS, Haney MJ, Zhao Y, Yuan D, Deygen I, Klyachko NL. et al. Engineering macrophage-derived exosomes for targeted paclitaxel delivery to pulmonary metastases: in vitro and in vivo evaluations. Nanomedicine. 2018;14:195-204
226. Haney MJ, Zhao Y, Jin YS, Li SM, Bago JR, Klyachko NL. et al. Macrophage-Derived Extracellular Vesicles as Drug Delivery Systems for Triple Negative Breast Cancer (TNBC) Therapy. J Neuroimmune Pharmacol. 2019
227. Rayamajhi S, Nguyen TDT, Marasini R, Aryal S. Macrophage-derived exosome-mimetic hybrid vesicles for tumor targeted drug delivery. Acta Biomater. 2019;94:482-94
228. Choi H, Kim Y, Mirzaaghasi A, Heo J, Kim YN, Shin JH. et al. Exosome-based delivery of super-repressor IkappaBalpha relieves sepsis-associated organ damage and mortality. Sci Adv. 2020;6:eaaz6980
229. Gangadaran P, Ahn BC. Extracellular Vesicle- and Extracellular Vesicle Mimetics-Based Drug Delivery Systems: New Perspectives, Challenges, and Clinical Developments. Pharmaceutics. 2020;12:E442
230. Ju S, Mu J, Dokland T, Zhuang X, Wang Q, Jiang H. et al. Grape exosome-like nanoparticles induce intestinal stem cells and protect mice from DSS-induced colitis. Mol Ther. 2013;21:1345-57
231. Wang Q, Ren Y, Mu J, Egilmez NK, Zhuang X, Deng Z. et al. Grapefruit-Derived Nanovectors Use an Activated Leukocyte Trafficking Pathway to Deliver Therapeutic Agents to Inflammatory Tumor Sites. Cancer Res. 2015;75:2520-9
232. Somiya M, Yoshioka Y, Ochiya T. Biocompatibility of highly purified bovine milk-derived extracellular vesicles. J Extracell Vesicles. 2018;7:1440132
233. Agrawal AK, Aqil F, Jeyabalan J, Spencer WA, Beck J, Gachuki BW. et al. Milk-derived exosomes for oral delivery of paclitaxel. Nanomedicine. 2017;13:1627-36
234. Sanwlani R, Fonseka P, Chitti SV, Mathivanan S. Milk-Derived Extracellular Vesicles in Inter-Organism, Cross-Species Communication and Drug Delivery. Proteomes. 2020;8:E11
235. Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367:eaau6977
236. Thery C, Witwer KW, Aikawa E, Alcaraz MJ, Anderson JD, Andriantsitohaina R. et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7:1535750
237. Huang G, Lin G, Zhu Y, Duan W, Jin D. Emerging technologies for profiling extracellular vesicle heterogeneity. Lab Chip. 2020;20:2423-2437
238. O'Brien K, Breyne K, Ughetto S, Laurent LC, Breakefield XO. RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat Rev Mol Cell Biol. 2020:1-22
239. Zhang H, Freitas D, Kim HS, Fabijanic K, Li Z, Chen H. et al. Identification of distinct nanoparticles and subsets of extracellular vesicles by asymmetric flow field-flow fractionation. Nat Cell Biol. 2018;20:332-43
240. Paolini L, Federici S, Consoli G, Arceri D, Radeghieri A, Alessandri I. et al. Fourier-transform Infrared (FT-IR) spectroscopy fingerprints subpopulations of extracellular vesicles of different sizes and cellular origin. J Extracell Vesicles. 2020;9:1741174
241. Kondratov K, Nikitin Y, Fedorov A, Kostareva A, Mikhailovskii V, Isakov D. et al. Heterogeneity of the nucleic acid repertoire of plasma extracellular vesicles demonstrated using high-sensitivity fluorescence-activated sorting. J Extracell Vesicles. 2020;9:1743139
242. Choi D, Go G, Kim DK, Lee J, Park SM, Di Vizio D. et al. Quantitative proteomic analysis of trypsin-treated extracellular vesicles to identify the real-vesicular proteins. J Extracell Vesicles. 2020;9:1757209
243. Wu X, Zhao H, Natalia A, Lim CZJ, Ho NRY, Ong CJ. et al. Exosome-templated nanoplasmonics for multiparametric molecular profiling. Sci Adv. 2020;6:eaba2556
244. Zhang P, Wu X, Gardashova G, Yang Y, Zhang Y, Xu L. et al. Molecular and functional extracellular vesicle analysis using nanopatterned microchips monitors tumor progression and metastasis. Sci Transl Med. 2020;12:eaaz2878
245. Sung BH, von Lersner A, Guerrero J, Krystofiak ES, Inman D, Pelletier R. et al. A live cell reporter of exosome secretion and uptake reveals pathfinding behavior of migrating cells. Nat Commun. 2020;11:2092
246. Wang J, Tu C, Zhang H, Zhang J, Feng Y, Deng Y. et al. Loading of metal isotope-containing intercalators for mass cytometry-based high-throughput quantitation of exosome uptake at the single-cell level. Biomaterials. 2020;255:120152
247. Kojima R, Bojar D, Rizzi G, Hamri GC, El-Baba MD, Saxena P. et al. Designer exosomes produced by implanted cells intracerebrally deliver therapeutic cargo for Parkinson's disease treatment. Nat Commun. 2018;9:1305
248. Sinha S, Hoshino D, Hong NH, Kirkbride KC, Grega-Larson NE, Seiki M. et al. Cortactin promotes exosome secretion by controlling branched actin dynamics. J Cell Biol. 2016;214:197-213
249. Gao J, Wang S, Wang Z. High yield, scalable and remotely drug-loaded neutrophil-derived extracellular vesicles (EVs) for anti-inflammation therapy. Biomaterials. 2017;135:62-73
250. Wang L, Abhange KK, Wen Y, Chen Y, Xue F, Wang G. et al. Preparation of Engineered Extracellular Vesicles Derived from Human Umbilical Cord Mesenchymal Stem Cells with Ultrasonication for Skin Rejuvenation. ACS Omega. 2019;4:22638-45
251. Lee D, Wee KW, Kim H, Han SI, Kwak S, Yoon DS. et al. Paper-Based Preconcentration and Isolation of Microvesicles and Exosomes. J Vis Exp. 2020 158
252. Fisher JD, Balmert SC, Zhang W, Schweizer R, Schnider JT, Komatsu C. et al. Treg-inducing microparticles promote donor-specific tolerance in experimental vascularized composite allotransplantation. Proc Natl Acad Sci U S A. 2019;116:25784-9
253. Zhang Y, Mei H, Chang X, Chen F, Zhu Y, Han X. Adipocyte-derived microvesicles from obese mice induce M1 macrophage phenotype through secreted miR-155. J Mol Cell Biol. 2016;8:505-17
Author contact
Corresponding author: Dr. Gang Wang, Department of Dermatology, Xijing Hospital, Fourth Military Medical University, 127 Changlexi Road, Xi'an 710032, China. E-mail: xjwgangedu.cn; Tel: 86-29-84775401.
 Global reach, higher impact
Global reach, higher impact