Impact Factor
Theranostics 2019; 9(25):7759-7771. doi:10.7150/thno.37574 This issue Cite
Review
STING activation in cancer immunotherapy
1. Department of Rehabilitation Medicine, Center for Translational Medicine, Precision Medicine Institute, The First Affiliated Hospital, Sun Yat-sen University, Guangzhou, 510080, China
2. Department of Pharmaceutics and Center for Pharmaceutical Engineering and Sciences, School of Pharmacy, Richmond, VA, 23298, USA
3. Massey Cancer Center, Virginia Commonwealth University, Richmond, VA, 23298, USA
4. Department of Radiation Oncology, Virginia Commonwealth University, Richmond, VA, 23298, USA
5. Department of Human and Molecular Genetics, Virginia Commonwealth University, Richmond, VA, 23298, USA.
6. Institute of Molecular Medicine, Virginia Commonwealth University, Richmond, VA, 23298, USA.
7. Institute for Structural Biology, Drug Discovery and Development, Virginia Commonwealth University, Richmond, VA, 23219, USA
*These authors contributed equally to this work
Abstract
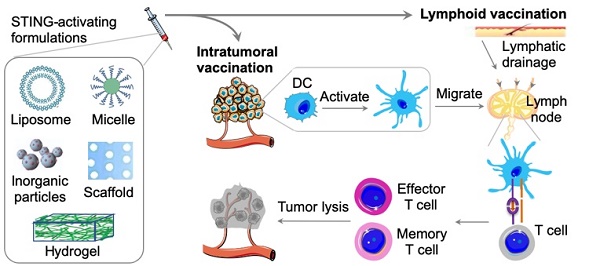
Cancer immunotherapy modulates and leverages the host immune system to treat cancer. The past decade has witnessed historical advancement of cancer immunotherapy. A myriad of approaches have been explored to elicit or augment anticancer innate immunity and/or adaptive immunity. Recently, activation of stimulator of interferon (IFN) genes (STING), an intracellular receptor residing in the endoplasmic reticulum, has shown great potential to enhance antitumor immunity through the induction of a variety of pro-inflammatory cytokines and chemokines, including type I IFNs. A number of natural and synthetic STING agonists have been discovered or developed, and tested in preclinical models and in the clinic for the immunotherapy of diseases such as cancer and infectious diseases. Cyclic dinucleotides (CDNs), such as cyclic dimeric guanosine monophosphate (c-di-GMP), cyclic dimeric adenosine monophosphate (c-di-AMP), and cyclic GMP-AMP (cGAMP), are a class of STING agonists that can elicit immune responses. However, natural CDNs are hydrophilic small molecules with negative charges and are susceptible to enzymatic degradation, leading to low bioavailability in target tissues yet unwanted toxicities and narrow therapeutic windows. Drug delivery systems, coupled with nucleic acid chemistry, have been exploited to address these challenges. Here, we will discuss the underlying immunological mechanisms and approaches to STING activation, with a focus on the delivery of STING agonists, for cancer immunotherapy.
Keywords: Stimulator of interferon genes (STING), cyclic dinucleotides, cyclic GMP-AMP synthase (cGAS), immunostimulatory adjuvants, drug delivery, cancer immunotherapy
 Global reach, higher impact
Global reach, higher impact