13.3
Impact Factor
Theranostics 2018; 8(22):6121-6131. doi:10.7150/thno.29070 This issue Cite
Research Paper
High performance immunochromatographic assay for simultaneous quantitative detection of multiplex cardiac markers based on magnetic nanobeads
1. Zhujiang Hospital, Southern Medical University, 253 Gongye Road, Guangzhou, Guangdong 510280, China.
2. Department of Instrument Science and Engineering, School of Electronic Information and Electrical Engineering, Shanghai Jiao Tong University, Shanghai Engineering Research Center for Intelligent diagnosis and treatment instrument, Key Laboratory of Thin Film and Microfabrication (Ministry of Education), Shanghai 200240, China.
3. Shanghai Institute of Microsystem and Information Technology, Chinese Academy of Sciences, Shanghai 200050, China.
4. School of Naval Architecture, Ocean & Civil Engineering, Shanghai Jiao Tong University, Shanghai 200240, China.
5. Lufeng People's Hospital, 43 Chengdong Avenue, Lufeng, Guangdong 516500, China.
Abstract
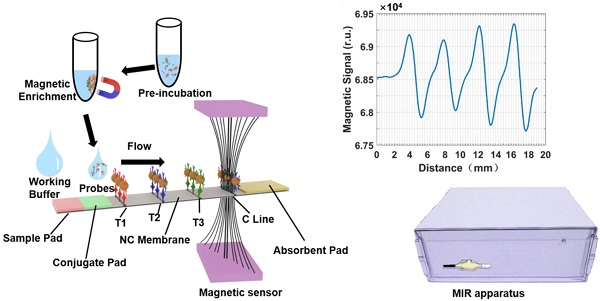
The detection of cardiac markers is critical to the diagnosis of acute myocardial infarction, and immunochromatographic assays are a common tool for point-of-care analysis.
Methods: We report a multiplexed lateral flow test strip for simultaneous quantitative detection of cardiac troponin I (cTnI), creatine kinase isoenzyme MB (CKMB), and myoglobin (Myo). Hydrophilic, monodisperse, stable, and carboxyl-modified (COOH-) magnetic nanobeads (MNBs) were used to construct immunomagnetic probes specific to the three cardiac markers. The detection area of the sandwich-style complexes contained three test lines for cTnI, CKMB, and Myo. The magnetic signal intensity of the detection area in the nitrocellulose membrane was measured via a magnetic immunochromatography reader developed in house.
Results: To optimize the assay, a modified working buffer was also investigated to improve the detection sensitivity, decrease the background noise, and shorten the detection time. The MNB-based immunochromatography test (MICT) strip offers a wide linear dynamic detection range, rapid detection, high sensitivity, and specificity. The limit of detection was 0.0089 ng/mL for cTnI, 0.063 ng/mL for CKMB, and 0.05 ng/mL for Myo with minimal cross-reactivity. There were 110 clinical human serum samples that were used to evaluate this platform with high correlation.
Conclusion: MICT shows great potential as a supplemental method for in vitro diagnostics in the laboratory or in other point-of-care testing (POCT) applications.
Keywords: magnetic nanobeads, immunochromatography, POCT, cardiac markers
 Global reach, higher impact
Global reach, higher impact