13.3
Impact Factor
Theranostics 2018; 8(20):5731-5743. doi:10.7150/thno.28782 This issue Cite
Research Paper
Spatial-Temporal Cellular Bioeffects from Acoustic Droplet Vaporization
Department of Biomedical Engineering and Environmental Sciences, National Tsing Hua University, Hsinchu, Taiwan
Abstract
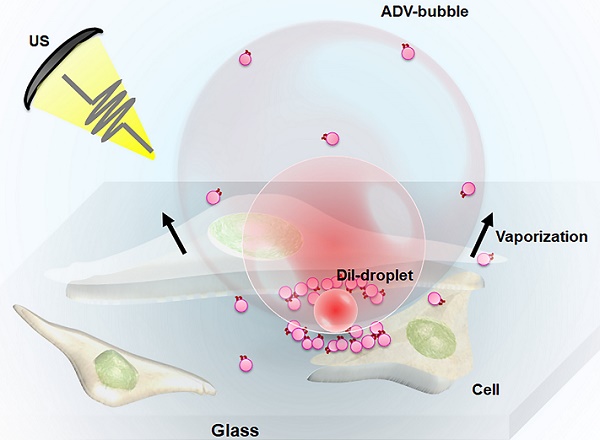
One of the major challenges in developing acoustic droplet vaporization (ADV)-associated therapy as an effective and safe strategy is the precise determination of the spatial cellular bioeffects after ADV (cell death or cell membrane permeabilization). In this study, we combined high-speed camera imaging and live-cell microscopic imaging to observe the transient dynamics of droplets during ADV and to evaluate the mechanical force on cells.
Methods: C6 glioma cells were co-incubated with DiI-labeled droplets (radius: 1.5, 2.25, and 3.0 μm). We used an acousto-optical system for high-speed bright-field (500 kfps) and fluorescence (40 kfps) microscopic imaging in order to visualize the dynamics of droplets under ultrasound excitation (frequency = 5 MHz, pressure = 5-8 MPa, cycle number = 3, pulse number = 1). Live-cell microscopic imaging was used to monitor the cell morphology, cell membrane permeabilization, and cell viability by membrane-anchored Lyn-yellow fluorescence protein, propidium Iodide staining, and calcein blue AM staining, respectively.
Results: We discovered that the spatial distribution of ADV-induced bioeffects could be mapped to the physical dynamics of droplet vaporization. For droplets with a 1.5 μm radius, the distance threshold for ADV-induced cell death (5.5±1.9 μm) and reversible membrane permeabilization (11.3±3.5 μm) was well correlated with the distance of ADV-bubble pressing downward to the floor (5.7±1.3 μm) and maximum distance of droplet expansion (11.5±2.6 μm), respectively. These distances were enlarged by increasing the droplet sizes and insonation acoustic pressures. The live-cell imaging results show that ADV-bubbles can directly disrupt the cell membrane layer and induce intensive intracellular substance leakage. Further, the droplets shed the payload onto nearby cells during ADV, suggesting ADV could directly induce adjacent cell death by physical force and enhancement of chemotherapy to distant cells.
Conclusion: This study provide new insights into the ADV-mediated physicochemical synergic effect for medical applications.
Keywords: acoustic droplets, ultrasound, acoustic droplet vaporization, cellular bioeffects, high-speed microscopy
 Global reach, higher impact
Global reach, higher impact