Impact Factor
Theranostics 2018; 8(20):5634-5644. doi:10.7150/thno.27776 This issue Cite
Research Paper
Engineered RNase P Ribozymes Effectively Inhibit the Infection of Murine Cytomegalovirus in Animals
1. Department of Biotechnology, College of Life Science and Technology, Jinan University, Guangzhou, Guangdong 510632, China
2. School of Public Health, University of California, Berkeley, CA 94720
3. School of Medicine, St. George's University, Grenada, West Indies
4. Taizhou Institute of Virology, Taizhou, Jiangsu 225300, China
5. Jiangsu Affynigen Biotechnolgies Inc, Taizhou, Jiangsu 225300, China
6. Guangzhou Qinheli Biotechnolgies Inc, Guangzhou, Guangdong 510600, China
7. School of Pharmacy, Shandong University of Traditional Chinese Medicine, Jinan, Shandong 250355, China
*These authors contribute equally to this study.
Abstract
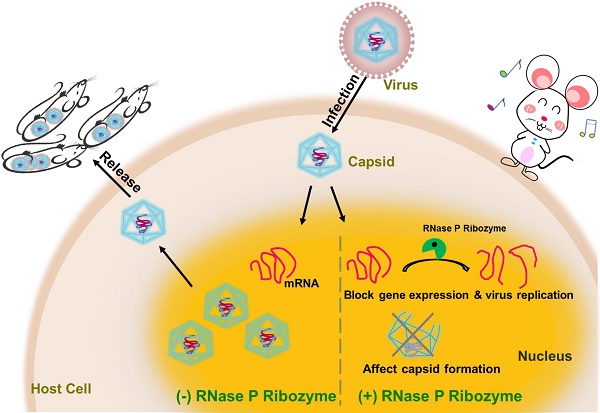
Rationales: Gene-targeting ribozymes represent promising nucleic acid-based gene interference agents for therapeutic application. We previously used an in vitro selection procedure to engineer novel RNase P-based ribozyme variants with enhanced targeting activity. However, it has not been reported whether these ribozyme variants also exhibit improved activity in blocking gene expression in animals.
Methods and Results: In this report, R388-AS, a new engineered ribozyme variant, was designed to target the mRNA of assemblin (AS) of murine cytomegalovirus (MCMV), which is essential for viral progeny production. Variant R338-AS cleaved AS mRNA sequence in vitro at least 200 times more efficiently than ribozyme M1-AS, which originated from the wild type RNase P catalytic RNA sequence. In cultured MCMV-infected cells, R338-AS exhibited better antiviral activity than M1-AS and decreased viral AS expression by 98-99% and virus production by 15,000 fold. In MCMV-infected mice, R388-AS was more active in inhibiting AS expression, blocking viral replication, and improving animal survival than M1-AS.
Conclusions: Our results provide the first direct evidence that novel engineered RNase P ribozyme variants with more active catalytic activity in vitro are also more effective in inhibiting viral gene expression in animals. Moreover, our studies imply the potential of engineering novel RNase P ribozyme variants with unique mutations to improve ribozyme activity for therapeutic application.
Keywords: antisense RNA, ribozyme, antiviral, gene targeting, gene therapy
 Global reach, higher impact
Global reach, higher impact