13.3
Impact Factor
Theranostics 2018; 8(19):5379-5399. doi:10.7150/thno.28391 This issue Cite
Review
V-ATPases and osteoclasts: ambiguous future of V-ATPases inhibitors in osteoporosis
1. State Key Laboratory of Military Stomatology, National Clinical Research Center for Oral Diseases, Department of Oral Biology, Clinic of Oral Rare and Genetic Diseases, School of Stomatology, the Fourth Military Medical University, 145 West Changle Road, Xi'an 710032, P. R. China.
2. Center for Genetic Epidemiology and Genomics, School of Public Health, Medical College of Soochow University, 199 Renai Road, Suzhou, Jiangsu, P. R. China.
3. Key Laboratory of Biomedical Information Engineering of Ministry of Education, and Institute of Molecular Genetics, School of Life Science and Technology, Xi'an Jiaotong University, 28 West Xianning Road, Xi'an 710049, People's Republic of China.
Abstract
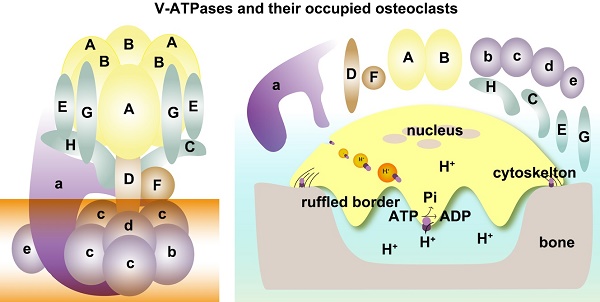
Vacuolar ATPases (V-ATPases) play a critical role in regulating extracellular acidification of osteoclasts and bone resorption. The deficiencies of subunit a3 and d2 of V-ATPases result in increased bone density in humans and mice. One of the traditional drug design strategies in treating osteoporosis is the use of subunit a3 inhibitor. Recent findings connect subunits H and G1 with decreased bone density. Given the controversial effects of ATPase subunits on bone density, there is a critical need to review the subunits of V-ATPase in osteoclasts and their functions in regulating osteoclasts and bone remodeling. In this review, we comprehensively address the following areas: information about all V-ATPase subunits and their isoforms; summary of V-ATPase subunits associated with human genetic diseases; V-ATPase subunits and osteopetrosis/osteoporosis; screening of all V-ATPase subunits variants in GEFOS data and in-house data; spectrum of V-ATPase subunits during osteoclastogenesis; direct and indirect roles of subunits of V-ATPases in osteoclasts; V-ATPase-associated signaling pathways in osteoclasts; interactions among V-ATPase subunits in osteoclasts; osteoclast-specific V-ATPase inhibitors; perspective of future inhibitors or activators targeting V-ATPase subunits in the treatment of osteoporosis.
Keywords: V-ATPase, osteoclasts, osteoporosis, osteopetrosis, pH, inhibitor, signaling pathways
 Global reach, higher impact
Global reach, higher impact