Impact Factor
Theranostics 2018; 8(18):5178-5199. doi:10.7150/thno.27798 This issue Cite
Research Paper
Targeted imaging and inhibition of triple-negative breast cancer metastases by a PDGFRβ aptamer
1. Istituto per l'Endocrinologia e l'Oncologia Sperimentale "G. Salvatore" (IEOS), CNR, Naples, Italy.
2. Istituto di Biostrutture e Bioimmagini, CNR, Naples, Italy.
3. Pathology Unit, Istituto Nazionale Tumori-IRCCS-Fondazione G. Pascale, Naples, Italy.
4. Functional Genomics, Istituto Nazionale Tumori-IRCCS-Fondazione G. Pascale, Naples, Italy.
Abstract
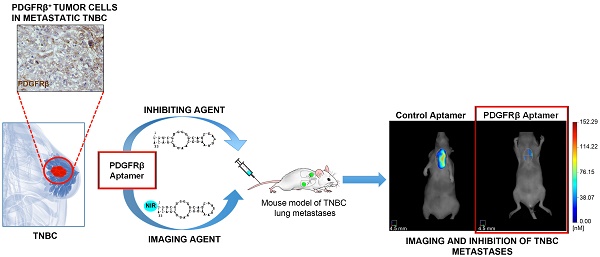
While the overall mortality for breast cancer has recently declined, management of triple-negative breast cancer (TNBC) is still challenging because of its aggressive clinical behavior and the lack of targeted therapies. Genomic profiling studies highlighted the high level of heterogeneity of this cancer, which comprises different subtypes with unique phenotypes and response to treatment. Platelet-derived growth factor receptor β (PDGFRβ) is an established mesenchymal/stem cell-specific marker in human glioblastoma and, as recently suggested, it may uniquely mark breast cancer cells with stem-like characteristics and/or that have undergone epithelial-mesenchymal transition.
Methods: Immunohistochemical analysis for PDGFRβ expression was performed on a human TNBC tissue microarray. Functional assays were conducted on mesenchymal-like TNBC cells to investigate the effect of a previously validated PDGFRβ aptamer on invasive cell growth in three-dimensional culture conditions, migration, invasion and tube formation. The aptamer was labeled with a near-infrared (NIR) dye and its binding specificity to PDGFRβ was assessed both in vitro (confocal microscopy and flow cytometry analyses) and in vivo (fluorescence molecular tomography in mice bearing TNBC xenografts). A mouse model of TNBC lung metastases formation was established and NIR-labeled PDGFRβ aptamer was used to detect lung metastases in mice untreated or intravenously injected with unlabeled aptamer.
Results: Here, we present novel data showing that tumor cell expression of PDGFRβ identifies a subgroup of mesenchymal tumors with invasive and stem-like phenotype, and propose a previously unappreciated role for PDGFRβ in driving TNBC cell invasiveness and metastases formation. We show that the PDGFRβ aptamer blocked invasive growth and migration/invasion of mesenchymal TNBC cell lines and prevented TNBC lung metastases formation. Further, upon NIR-labeling, the aptamer specifically bound to TNBC xenografts and detected lung metastases.
Conclusions: We propose PDGFRβ as a reliable biomarker of a subgroup of mesenchymal TNBCs with invasive and stem-like phenotype as well as the use of the PDGFRβ aptamer as a high efficacious tool for imaging and suppression of TNBC lung metastases. This study will allow for the significant expansion of the current repertoire of strategies for managing patients with more aggressive TNBC.
Keywords: aptamer, PDGFRβ, invasiveness, mesenchymal TNBC subtype, metastases.
 Global reach, higher impact
Global reach, higher impact