13.3
Impact Factor
Theranostics 2018; 8(13):3707-3721. doi:10.7150/thno.25166 This issue Cite
Research Paper
AP-2β inhibits hepatocellular carcinoma invasion and metastasis through Slug and Snail to suppress epithelial-mesenchymal transition
1. Key Laboratory of Protein Chemistry and Development Biology of State Education Ministry of China, College of Life Science, Hunan Normal University, Changsha, 410081, P.R. China
2. Department of Endocrinology, Endocrinology Research Center, Xiangya Hospital of Central South University, Changsha, 410011, P.R. China
3. State Key Laboratory of Developmental Biology of Freshwater Fish, College of Life Science, Hunan Normal University, Changsha, 410081, P.R. China
4. College of Engineering and Design, Hunan Normal University, Changsha, 410081, P.R. China
5. The National & Local Joint Engineering Laboratory of Animal Peptide Drug development, College of Life Science, Hunan Normal University, Changsha, 410081, P.R. China
Abstract
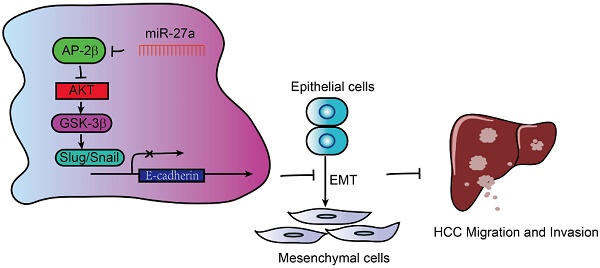
Transcription factor AP-2β plays an important role in human cancer, but its clinical significance in hepatocellular carcinogenesis is largely unknown.
Methods: AP-2β expression was detected in human hepatocellular cancer (HCC) tissues and cell lines. The effects of AP-2β on HCC proliferation, migration, invasion, tumor formation and metastasis were evaluated by MTT, colony formation and transwell assays in vitro and mouse experiments in vivo. The association between AP-2β and miR-27a/EMT markers in HCC cell lines and tissues was analyzed.
Results: AP-2β expression was decreased in HCC tissues and cell lines. Reduced expression of AP-2β was significantly associated with more advanced tumor stages and larger tumor sizes. The overexpression of AP-2β reduced HCC proliferation, migration, invasion, tumor formation and metastasis in vitro and in vivo. Additionally, AP-2β overexpression increased the sensitivity of HCC cells to cisplatin. Moreover, AP-2β modulates the levels of EMT markers through Slug and Snail in HCC cell lines and tissues. Furthermore, oncogenic miR-27a inhibits AP-2β expression by binding to the AP-2β 3′ untranslated region (UTR) and reverses the tumor suppressive role of AP-2β.
Conclusion: These results suggested that AP-2β is lowly expressed in HCC by inhibiting EMT signaling to regulate HCC cell growth and migration. Therefore, AP-2β in the novel miR-27a/AP-2β/Slug/EMT regulatory axis enhances the chemotherapeutic drug sensitivity of HCC and might represent a potential target for evaluating the treatment and prognosis of human HCC.
Keywords: AP-2β, hepatocellular carcinoma, epithelial-to-mesenchymal transition (EMT), Slug, miR-27a
 Global reach, higher impact
Global reach, higher impact