13.3
Impact Factor
Theranostics 2018; 8(12):3176-3188. doi:10.7150/thno.23207 This issue Cite
Research Paper
The HER2 inhibitor lapatinib potentiates doxorubicin-induced cardiotoxicity through iNOS signaling
1. Institute of Biomedical Sciences, Academia Sinica, Taiwan;
2. School of Pharmacy, National Taiwan University, Taiwan;
3. Department of Oncology, National Taiwan University and Hospital, Taiwan;
4. Institute of Medical Genomics and Proteomics, National Taiwan University College of Medicine, Taiwan;
5. Institute of Clinical Medicine, National Taiwan University College of Medicine, Taiwan;
6. Division of Cardiovascular Surgery, Department of Surgery, National Taiwan University Hospital, Taiwan.
Abstract
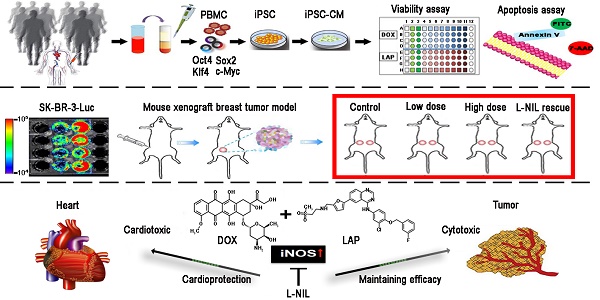
Rationale: Lapatinib (LAP) is a crucial alternative to trastuzumab upon the onset of drug resistance during treatment of metastatic human epidermal growth factor receptor 2-positive breast cancer. Like trastuzumab, LAP is commonly used alongside anthracyclines as a combination therapy, due to enhanced anti-cancer efficacy. However, this is notably associated with cardiotoxicity so it is imperative to understand the mechanisms driving this cardiotoxicity and develop cardioprotective strategies. To this end, here we utilize human pluripotent stem cell-derived cardiomyocytes (hPSC-CMs), which exhibit several characteristics representative of in vivo cardiomyocytes that make them breakthrough models to study drug toxicity.
Methods: We investigated LAP- and doxorubicin (DOX)-induced toxicity in hPSC-CMs and evaluated the involvement of inducible nitric oxide (NO) synthase (iNOS). The significance of iNOS-mediated cardiotoxicity was furthermore evaluated in animal studies.
Results: LAP synergistically increased DOX toxicity in hPSC-CMs in a dose- and time-dependent manner. At concentrations that were otherwise non-apoptotic when administered separately, LAP significantly potentiated DOX-induced hPSC-CM apoptosis. This was accompanied by increased iNOS expression and pronounced production of NO. iNOS inhibition significantly reduced hPSC-CM sensitivity to LAP and DOX co-treatment (LAP-plus-DOX), leading to reduced apoptosis. Consistent with our observations in vitro, delivery of an iNOS inhibitor in mice treated with LAP-plus-DOX attenuated myocardial apoptosis and systolic dysfunction. Moreover, inhibition of iNOS did not compromise the anti-cancer potency of LAP-plus-DOX in a murine breast cancer xenograft model.
Conclusions: Our findings suggest that iNOS inhibition is a promising cardioprotective strategy to accompany HER2-inhibitor/anthracycline combination therapies. Furthermore, these results support the promise of hPSC-CMs as a platform for investigating cardiotoxicity and developing cardioprotectants as a whole.
Keywords: HER2 inhibitor, cardiotoxicity, doxorubicin, iPSC, iNOS
 Global reach, higher impact
Global reach, higher impact