Impact Factor
Theranostics 2017; 7(17):4135-4148. doi:10.7150/thno.20955 This issue Cite
Research Paper
Tethering Interleukin-22 to Apolipoprotein A-I Ameliorates Mice from Acetaminophen-induced Liver Injury
Department of Microbiological and Biochemical Pharmacy & The Key Laboratory of Smart Drug Delivery, Ministry of Education, School of Pharmacy, Fudan University, 826 Zhangheng Road, Shanghai, 201203, P. R. China.
* These authors contributed equally to this work.
Abstract
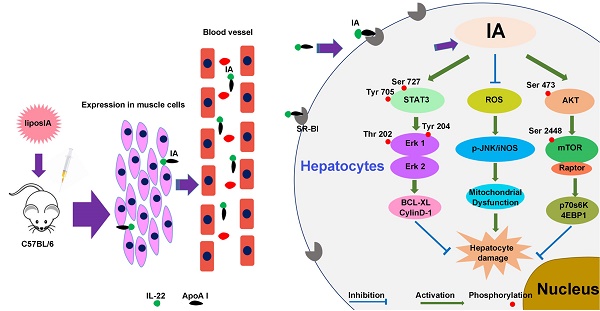
Increasing evidence indicates that interleukin-22 (IL-22) holds tremendous potential as a protective agent in preventing liver injury, but its pleiotropic effects and pathogenic role in carcinogenesis, rheumatoid arthritis and psoriasis restrict its systemic application. Here, we first developed a nanoparticle (liposIA) as a liver-targeted agent through IL-22 tethered to apolipoprotein A-I (ApoA-I) in a gene therapy vector. LiposIA was prepared using thin film dispersion method and the complexes exhibited desirable nanoparticle size, fine polydisperse index, highly efficient transfection, and excellent serum and storage stability. Biodistribution and hepatic STAT3 phosphorylation studies revealed that IL-22 tethered to ApoA-I led to highly efficient liver targeting. More importantly, our studies showed that a single-dose of liposIA was able to protect mice against acetaminophen-induced liver injury and did not initiate inflammatory response or systemic toxicity in vivo. During this process, activated STAT3/Erk and Akt/mTOR signaling transductions were observed, as well as inhibition of reactive oxygen species (ROS) generation, which prevented mitochondrial dysfunction. These studies demonstrated that IL-22 tethered to apolipoprotein A-I could target and ameliorate acetaminophen-induced acute liver injury, which highlighted that a targeted strategy for IL-22 delivery might have broad utility for the protection of hepatocellular damage.
Keywords: apolipoprotein A-I, interleukin-22, liver targeting, acetaminophen-induced acute liver injury.
 Global reach, higher impact
Global reach, higher impact