Impact Factor
Theranostics 2017; 7(17):4099-4117. doi:10.7150/thno.20730 This issue Cite
Research Paper
Vascularized Tissue-Engineered Model for Studying Drug Resistance in Neuroblastoma
1. Laboratory for Stem Cells and Tissue Engineering, Department of Biomedical Engineering, Columbia University, New York, NY, USA;
2. Institute of Advanced Biomedical Engineering and Science, Tokyo Women's Medical University, TWIns, Tokyo, Japan;
3. Faculty of Science and Engineering, Waseda University, TWIns, Tokyo, Japan;
4. Department of Pediatrics, Memorial Sloan Kettering Cancer Center, New York, NY. USA;
5. Department of Medicine, Columbia University, New York, NY, USA.
Abstract
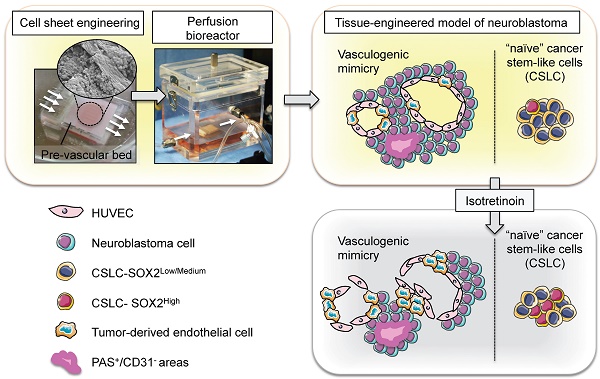
Neuroblastoma is a vascularized pediatric tumor derived from neural crest stem cells that displays vasculogenic mimicry and can express a number of stemness markers, such as SOX2 and NANOG. Tumor relapse is the major cause of succumbing to this disease, and properties attributed to cancer stem-like cells (CSLC), such as drug-resistance and cell plasticity, seem to be the key mechanisms. However, the lack of controllable models that recapitulate the features of human neuroblastoma limits our understanding of the process and impedes the development of new therapies. In response to these limitations, we engineered a perfusable, vascularized in vitro model of three-dimensional human neuroblastoma to study the effects of retinoid therapy on tumor vasculature and drug-resistance.
METHODS: The in vitro model of neuroblastoma was generated using cell-sheet engineering and cultured in a perfusion bioreactor. Firstly, we stacked three cell sheets containing SKNBE(2) neuroblastoma cells and HUVEC. Then, a vascular bed made of fibrin, collagen I and HUVEC cells was placed onto a collagen-gel base with 8 microchannels. After gelling, the stacked cell sheets were placed on the vascular bed and cultured in the perfusion bioreactor (perfusion rate: 0.5 mL/min) for 4 days. Neuroblastoma models were treated with 10μM isotretionin in single daily doses for 5 days.
RESULTS: The bioengineered model recapitulated vasculogenic mimicry (vessel-like structure formation and tumor-derived endothelial cells-TECs), and contained CSLC expressing SOX2 and NANOG. Treatment with Isotretinoin destabilized vascular networks but failed to target vasculogenic mimicry and augmented populations of CSLCs expressing high levels of SOX2. Our results suggest that CSLCs can transdifferentiate into drug resistant CD31+-TECs, and reveal the presence of an intermediate state STEC (stem tumor-derived endothelial cell) expressing both SOX2 and CD31.
CONCLUSION: Our results reveal some roles of SOX2 in drug resistance and tumor relapse, and suggest that SOX2 could be a therapeutic target in neuroblastoma.
Keywords: Neuroblastoma, Bioengineering, perfusion bioreactor, vasculogenic mimicry, SOX2, cell plasticity.
 Global reach, higher impact
Global reach, higher impact