Impact Factor
Theranostics 2026; 16(11):5816-5829. doi:10.7150/thno.132930 This issue Cite
Review
Future opportunities and nuances with the use of PSMA PET in prostate cancer (MD PET 1)
1. Department of Medical Oncology, Dana-Farber Cancer Institute, Boston, MA, USA.
2. U.S. Urology Partners and Associated Medical Professionals of New York, Syracuse, NY, USA.
3. Fred Hutchinson Cancer Center and University of Washington, Seattle, WA, USA.
4. Memorial Sloan Kettering Cancer Center, New York, NY, USA.
5. Department of Urology, Thomas Jefferson University, Philadelphia, PA, USA.
6. Department of Radiology, Division of Nuclear Medicine, Saint Louis University Hospital, St. Louis, MO, USA.
7. Prostate Cancer Foundation, Santa Monica, CA, USA.
8. Center of Advanced Radiation Excellence (CARE) and Radiation Oncology Research, Associated Medical Professionals / US Urology Partners, Syracuse, NY, USA.
Received 2026-2-10; Accepted 2026-3-21; Published 2026-4-9
Abstract
Imaging with prostate-specific membrane antigen (PSMA) PET has significantly improved prostate cancer staging with superior diagnostic performance compared to conventional methods. Although it is increasingly adopted in clinical practice, several barriers hinder its full integration into routine workflows. This review highlights the existing knowledge gaps, infrastructure limitations, and inconsistencies in interpretation that affect the utility of PSMA PET across healthcare settings. We examine the potential reasons behind variability in scan performance, including scanner design, detector technology, sensitivity, and resolution, as well as the accreditation status of the facilities and reader expertise. We also highlight the inconsistent understanding of PSMA PET ordering practices, particularly among urologists, and the influence of ownership-driven utilization, both of which contribute to underuse and overuse. Radiology reporting that lacks sufficient detail and a shortage of trained nuclear medicine specialists may present additional challenges to effective treatment planning, although diagnostic radiologists also contribute to PET scan interpretation. This review highlights the potential role of standardized reporting protocols, accreditation, expanded education, and integration of AI tools in enhancing diagnostic accuracy and consistency. Additionally, we examine the impact of PSMA PET on clinical decision-making in unfavorable intermediate-, high risk-, and biochemically recurrent prostate cancer, as well as the emerging role of PSMA PET-derived metrics in staging, biopsy guidance, and treatment planning. While PSMA PET has shown value in modifying management strategies, its clinical benefit requires validation through future, prospective, outcome-driven studies. In addition, emerging applications of PSMA PET in non-prostate malignancies hold the potential to transform diagnostic and therapeutic approaches beyond prostate cancer.
Keywords: MD PET, prostate cancer, prostate-specific membrane antigen, PSMA-PET/CT, clinical decision-making, diagnostic imaging
Introduction
Prostate cancer, the second most common cancer in men, accounts for approximately 7.3% of the global cancer burden [1]. In the United States, it represents 15.4% of all new cancer diagnoses, with an estimated 313,830 new cases and 36,320 deaths projected for 2026 [2]. The 5-year relative survival rate is nearly 100% for localized and regional disease but declines to 37.9% for metastatic prostate cancer [3]. From a therapeutic point of view, clinically important improvements in survival from addition of abiraterone to ADT have been reported to be maintained for longer than 7 years in very high risk localized prostate cancer either with positive nodes or 2 of 3 criteria being Gleason 8-10, PSA>40 ng/mL and/or T3-4 status. [4]. Therefore, early and accurate staging is crucial for determining therapeutic strategies and reducing prostate cancer-specific mortality. Imaging plays a pivotal role from diagnosis and staging to the detection of recurrence and confirming eligibility of patients prior to prostate-specific membrane antigen (PSMA)-targeted radioligand therapy. There is also significant interest in integrating PSMA PET image-based information from radiomics into a multiomics framework, with the aim to combine biomolecular-level information with imaging data. The clinical application of radiomics can help to identify the relationship between imaging results and the clinical outcome of interest [5].
Historically, conventional imaging modalities such as computed tomography (CT) and magnetic resonance imaging (MRI) have had limitations in detecting small lymph node metastases, distant metastases, as well as early recurrence [6-8]. Bone scintigraphy, although commonly used to detect bone metastases, has a high false-positive rate, which can lead to overstaging of disease [8]. These diagnostic pitfalls underscored the need for more sensitive and specific imaging techniques, which can detect previously occult disease with greater accuracy and at the earlier stages of disease progression. The emergence of PSMA positron emission tomography (PET) has helped address many of these challenges [9-11]. Notably, it enables the detection of PSMA-positive lesions in patients with negative conventional imaging, underscoring the diagnostic value of PSMA-targeted molecular imaging [12].
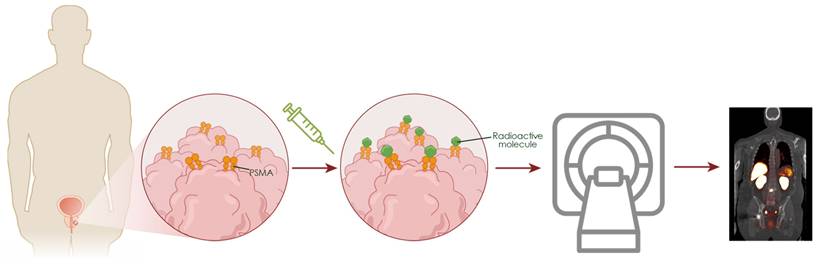
PSMA is a transmembrane glycoprotein markedly overexpressed in prostate cancer cells compared to normal tissues, making it an ideal target for molecular imaging [13,14]. PSMA PET tracers radiolabeled with isotopes such as gallium-68 (68Ga) or fluorine-18 (18F), bind specifically to the extracellular domain of PSMA and undergo internalization via endocytosis. This accumulation enables high-contrast imaging of PSMA-expressing lesions (Figure 1). Currently, the U.S. Food and Drug Administration (FDA)-approved PSMA PET agents include piflufolastat F 18 (PYLARIFY, PYLARIFY TRUVU), flotufolastat F 18 (POSLUMA), and 68Ga-PSMA-11 (LOCAMETZ, ILLUCCIX, GOZELLIX) [15-20]. All are indicated for prostate cancer patients with suspected metastasis who are candidates for initial definitive therapy, or with suspected recurrence based on a rising serum prostate-specific antigen (PSA) level. In addition, the use of PSMA PET agents for patient selection for men with metastatic prostate cancer who are candidates for PSMA-targeted radioligand therapy with 177Lu-vipivotide tetraxetan (PLUVICTO), is consistent with current PLUVICTO FDA labeling and National Comprehensive Cancer Network (NCCN) guidelines [21,22] (Table 1).
Imaging with PSMA PET. PSMA is a transmembrane glycoprotein overexpressed on the surface of prostate cancer cells compared to normal tissues. When PSMA PET radiotracers are injected into the bloodstream they bind PSMA, allowing for specific detection of prostate tumors/metastases through PET.
Overview of Commercially Available, FDA-Approved PSMA PET Agents
| Product Name | Imaging Agent | Half-Life | Approval Year | Indications & NCCN Guidelines* | Pivotal Studies | Diagnostic Performance Summary |
|---|---|---|---|---|---|---|
| PYLARIFY | Piflufolastat F 18 | 110 minutes | 2021 | Initial staging; Suspected recurrence based on elevated PSA; Selection for radioligand therapy with 177Lu-vipivotide tetraxetan | CONDOR, OSPREY | CONDOR (BCR) [12,71] CLR: 84.8-87.0% Detection rate: 59-66% OSPREY (Initial Staging - Cohort A) [7,12] Specificity 96% PPV 77% Suspected recurrence/metastases (Cohort B) Sensitivity 95.8% PPV 81.9% |
| ILLUCCIX | 68Ga-PSMA-11 (68Ga-gozetotide) | 68 minutes | 2021 | Initial staging; Suspected recurrence based on elevated PSA; Selection for radioligand therapy with 177Lu-vipivotide tetraxetan | PSMA-PreRP, PSMA-BCR, VISION | PSMA-BCR [14] Detection rate: 54% (95% CI: 50-57%) PSMA-PreRP Sensitivity 47% Specificity 90% VISION: 87% of patients screened met PSMA-positive imaging criteria for therapy selection |
| LOCAMETZ | 68Ga-PSMA-11 (68Ga-gozetotide) | 68 minutes | 2022 | Initial staging; Suspected recurrence based on elevated PSA; Selection for radioligand therapy with 177Lu-vipivotide tetraxetan | PSMA-PreRP, PSMA-BCR, VISION | Same as ILLUCCIX [15] |
| POSLUMA | Flotufolastat F 18 | 110 minutes | 2023 | Initial staging; Suspected recurrence based on rising PSA; Selection for radioligand therapy with 177Lu-vipivotide tetraxetan | LIGHTHOUSE SPOTLIGHT | LIGHTHOUSE (Initial Staging) [13] Sensitivity: 23-30% Specificity: 93-97% SPOTLIGHT (Suspected Recurrence) Detection rate: 83% |
| GOZELLIX | 68Ga-PSMA-11 (68Ga-gozetotide) | 68 minutes | 2025 | Initial staging; Suspected recurrence based on elevated PSA; Selection for radioligand therapy with 177Lu-vipivotide tetraxetan | PSMA-PreRP, PSMA-BCR | PSMA-BCR [16] Detection rate: 54% (95% CI: 50-57%) PSMA-PreRP Sensitivity 47% Specificity 90% |
*As per updated FDA labeling for 177Lu-vipivotide tetraxetan and NCCN guidelines, all FDA-approved PSMA PET agents may be used for patient selection. This indication is not included in the original prescribing information for PYLARIFY, POSLUMA, or GOZELLIX.
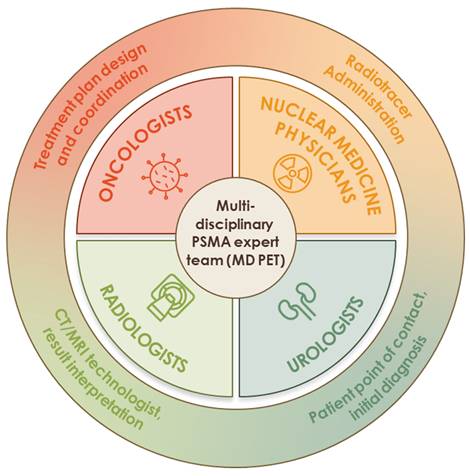
PSMA PET agents have significantly changed the diagnostic landscape of prostate cancer by enabling more accurate staging, restaging, and treatment planning [23,24]. This article, which brings together a Multi-Disciplinary PSMA Expert Team (MD PET) working group (Figure 2), provides a unique perspective that highlights the unmet need and knowledge gaps in the clinical application of PSMA PET in prostate cancer and summarizes ongoing trials aimed at addressing these gaps. It also provides strategies to overcome current challenges while exploring new opportunities to enhance the clinical utility of PSMA PET.
The multi-disciplinary PSMA expert team (MD PET). The multi-disciplinary PSMA expert team (MD PET) consists of 4 core disciplines: oncology, urology, nuclear medicine, and radiology. Multidisciplinary care may improve patient-specific decision-making.
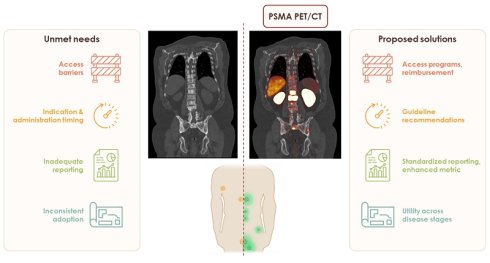
Access Barriers to PSMA PET Uptake and Use: Overview of the Unmet Need
Infrastructure barriers
The availability of imaging infrastructure significantly influences the utilization of PSMA PET. Fixed, onsite PET scanners that are typically housed within large academic or specialty centers require substantial capital and ongoing operational investment [25] and limited numbers and locations contribute to long wait times. As a result, access remains limited across many community healthcare settings.
Coverage barriers
Coverage is another key barrier to PSMA PET access, influenced by both the patient population and the policies of individual insurance providers. For example, among Veteran Affairs (VA) populations, access to PSMA PET appears to be less restricted with a small percentage of scans ordered off-label for low- and favorable intermediate-risk prostate cancer cases [26]. In contrast, non-VA populations can face more challenges due to variability in payer policies [27]. Moreover, the choice of PSMA PET agent is often determined by the insurance provider [28]. Though the NCCN guidelines support the use of any FDA-approved PSMA PET agent for patient selection prior to 177Lu-PSMA-617 therapy, factors such as pricing beyond the initial three-year coverage period may influence agent choice in clinical practice. These payer-driven constraints may limit optimal agent selection based on patient-specific clinical scenarios. Furthermore, the issues of insurance and reimbursement barriers are not limited to the US. In both Europe and Asia, due to its high cost, PSMA PET is not always covered thus requiring patients to bear the out-of-pocket cost of imaging related expenses or lose access altogether [29,30].
Addressing this Unmet Need
Infrastructure expansion through funding and access programs
Investment in PET infrastructure, including both fixed and mobile units [31], may be facilitated through public-private partnerships, federal funding initiatives, or value-based healthcare models [32]. Expanded access programs, such as those sponsored by the Department of Veterans Affairs or National Cancer Institute-designated cancer centers, may improve equity in imaging access across both urban and rural settings [33].
Accreditation programs offered by professional societies such as the Society of Nuclear Medicine and Molecular Imaging (SNMMI) and the American College of Radiology promote standardized acquisition protocols for PSMA PET/CT [23]. Facilities accredited under these programs ensure high-quality imaging and minimize scan variability across sites.
Insurance reform
Payer-driven restrictions may delay diagnosis or limit patient access to necessary care [27,28]. Reimbursement policy changes are needed to minimize coverage-related variability across providers and improve accessibility to PSMA PET. Advocacy from clinical societies and real-world data demonstrating clinical utility plays a crucial role in mediating meaningful changes in the policy.
Cost-effectiveness evidence
Beyond infrastructure expansion and insurance reform, studies have been conducted to determine whether the use of PSMA PET for prostate cancer is cost-effective. An analysis performed by Privé et al. reported that for PI-RADS 3 lesions, PSMA-PET/CT was marginally cost-effective, improving per-patient Quality-Adjusted Life Years (QALY) by 0.002 [34]. Separately, Yee et al. reported that PSMA PET with piflufolastat F 18 was a cost-effective option for men with prostate cancer in the US, with higher associated life-years, QALY, and greater net monetary benefit than non-PSMA-targeted imaging agents [35]. While more pharmacoeconomic and outcomes-based studies are needed, preliminary evidence suggests that PSMA PET can be cost effective for some prostate cancer patients.
Inconsistent Understanding on When to Order a PSMA PET Scan: Overview of the Unmet Need
Although there is an increase in the adoption of PSMA PET, clinician ordering patterns vary considerably in terms of timing and indication for scans [36]. This could be due to evolving guidelines, limited imaging experience, or the biological heterogeneity of the disease itself. Such inconsistency in PET ordering practices could lead to either over or underutilization of the PSMA PET, potentially affecting disease staging and treatment planning. Multidisciplinary care involving urologists, radiation oncologists, medical oncologists, and nuclear medicine colleagues, when available, may improve patient-specific decision-making [37].
Addressing this unmet need
Clinical guidelines have emphasized the appropriate integration of PSMA PET in prostate cancer staging and treatment planning [22-24]. The 2026 NCCN guidelines recommend the use of PSMA imaging in the initial staging of patients with unfavorable intermediate- and high-risk prostate cancer as well as in patients with biochemical recurrence following curative-intent therapy [22]. Additionally, PSMA PET is recommended for patients with metastatic castration-resistant prostate cancer (mCRPC) when assessing eligibility for PSMA-targeted radioligand therapy. The NCCN recognizes the superior sensitivity and specificity of PSMA PET tracers compared to conventional imaging modalities, such as CT, MRI, and bone scan, in detecting disease at both initial staging and biochemical recurrence. The panel further notes that conventional imaging is not a necessary prerequisite for PSMA PET and that PSMA PET/CT or PSMA PET/MRI can serve as an equally effective front-line imaging tool [22]. Although 68Ga-PSMA-11 is the only agent formally approved for selecting patients for 177Lu-PSMA-617 radioligand therapy, the NCCN panel acknowledges that piflufolastat F 18 and flotufolastat F 18 may also be used based on their comparable imaging performance [22]. Furthermore the U.S. prescribing information for 177Lu-PSMA-617 for patients with PSMA-positive mCRPC prior to chemotherapy is now agnostic to the choice of PSMA PET agent for patient selection only requiring that the PET product be FDA approved [21].
Knowledge Gaps When Conducting and Interpreting PSMA PET: Overview of the Unmet Need
Variability in scan performance across sites
The performance of PSMA PET scans varies across different clinical settings (e.g., academic institutions, freestanding outpatient centers, etc.) due to differences in scanner technology, accreditation status, reader expertise, as well as variability in imaging protocols and imaged field of view among FDA-approved PSMA PET agents. PET/CT scanners vary in design with differences in detector technology, sensitivity, resolution, and reconstruction algorithms impacting overall performance [38-41]. Studies comparing digital PET to conventional analog PET have demonstrated variations in standardized uptake value (SUV) measurements and improved detection of small lesions with digital platforms [40,41].
Reader variability also contributes to inconsistencies in image interpretation. Interobserver differences in PSMA expression scoring and lesion classification are well documented and influenced by readers' training and experience [42,43]. Nuanced clinical knowledge of different disease states and their associated imaging requirements (e.g., initial staging, biochemical recurrence [BCR], mCRPC) is crucial for accurately interpreting PSMA PET scans in various scenarios. While academic centers may possess experienced readers, the equipment may be antiquated in some facilities. In some clinical trials, sites are ineligible to participate if the PET scanner is older than 10 years; however, such cutoffs do not exist in real-world settings. Therefore, some PET centers may continue to use outdated scanners. Conversely, freestanding outpatient centers are often less likely to be accredited and may utilize less advanced scanners. These centers tend to get reimbursed less and often shorten the imaging protocols to improve patient throughput. Additionally, technical factors such as the choice of radiotracer, urinary bladder volume, and acquisition timing can influence image quality [23,44-46].
Inadequate reporting
Radiology reports often lack the level of detail necessary for effective treatment planning. Reports that fail to specify lesion location or omit image references may leave referring clinicians with insufficient information for decision-making [47]. Additionally, fused images stored in picture archiving and communication systems (PACS) are often difficult to interpret. Without access to advanced software many clinicians are unable to adjust the images to locate described lesions or confirm findings, limiting the clinical utility of the scan. Furthermore, imaging metrics such as whole-body SUVmean have been shown to predict outcomes in the VISION and TheraP trials [48,49]. However, no standard reporting or nuclear medicine reports present such information. Incorporating such metrics into routine reporting could enhance clinical decision-making and is possible with artificial intelligence (AI) integration.
Shortage of trained nuclear medicine physicians
There is a shortage of trained nuclear medicine physicians in the United States [50]. While radiologists are permitted to interpret scans, they are not board-certified in nuclear medicine, and interpreting complex scans requires extensive experience. The number of Accreditation Council for Graduate Medical Education (ACGME)-certified nuclear medicine training has declined significantly over the years while demand has increased, widening the gap [50]. This shortage of trained experts also presents a challenge for implementing AI, as it is difficult to account for all the variables involved in scan interpretation [51].
Addressing this unmet need
Optimizing performance and reading of scans
The optimal image quality depends on standardized scan acquisition protocols. Factors, such as the injection process, acquisition time, and bladder volume, influence image quality [22,52]. Since different tracers may exhibit distinct uptake and biodistribution patterns, the choice of radiotracer directly affects scan performance [53]. Optimizing injection protocols, including timing, dosage, and administration, enhances lesion detectability while minimizing artifacts [23,54]. While the typical imaging time for a PSMA PET scan is 60 minutes post-injection, delayed imaging has shown potential for detecting additional metastatic sites, particularly in cases of small-volume disease [23,55,56]. Longer acquisition time improves image quality by increasing detectability and reducing noise [46].
Urinary bladder volume is also an important factor, as high tracer accumulation can create artifacts that blur images and obscure pelvic lesions [23,45]. This interference is especially relevant for PSMA-ligands with kidney-dominant excretion, where residual activity in the urinary system may lead to false positive or negative findings. Strategies such as pre-scan hyperhydration, bladder voiding, and use of diuretics are commonly recommended to reduce urinary tracer interference and improve scan quality [23]. Additionally, selecting tracers with lower urinary excretion may further minimize bladder-related artifacts [45,57]. However, well controlled, prospective head-to-head comparative studies are needed to validate whether this approach improves diagnostic accuracy.
Multicenter studies require standardized imaging protocols to ensure reproducibility across institutions. Without direct head-to-head trials comparing different imaging agents, interpreting scans with varying tracers remains a challenge.
How to avoid interpretive pitfalls
With the challenges of false positives and tracer-related variability, there are a number of approaches to mitigate these potential issues. First, having a familiarity with typical lesions unrelated to prostate cancer can help with the interpretation of false positives. For instance, benign findings such as granulomatous prostatitis in the prostate, fibrous dysplasia and Paget's disease in bone, and desmoid tumors in soft tissue can be properly contextualized to avoid misinterpretation. Another example is modest PSMA uptake in the inguinal lymph nodes which is a common observation but typically does not suggest pathologic involvement. Isolated metastatic rib lesions are also often difficult to distinguish from a healing fracture, and it is well known that other tumor types can appear on PSMA PET. Lastly, standardized reporting systems such as PSMA-RADS may help to address issues related to reader variability [58].
Enhancing report utility
Imaging reports play a crucial role in guiding treatment decisions [23,47]. The utility of the report can be enhanced by providing the target site and image slice to help guide the reader. Community clinicians may benefit from reports that in addition to summarizing the findings, provide educational insights into technical aspects as well as guidance on scan interpretation. Standardized reporting of PSMA PET can improve communication between imaging specialists and referring physicians, ensuring that key findings are conveyed clearly and consistently [47].
Potential of AI to support interpretation
AI-driven tools can enhance reader ability by providing quantification of key prognostic and predictive metrics, such as total tumor volume and whole-body SUVmean [59]. The aPROMISE platform is designed to standardize tumor burden evaluation with piflufolastat F 18 PET/CT and represents a significant advancement in prostate cancer imaging [60]. Compared to conventional imaging, aPROMISE-assisted reads significantly upstaged patients with localized and regional prostate tumors and reduced inter-reader variability [60]. Johnsson et al. reported that aPROMISE effectively automates anatomic contextualization, lesion detection, and SUV-based quantification, thereby supporting standardized reporting of PSMA PET/CT in clinical practice [61]. Recently, a study has reported high agreement between automated (aPROMISE) and semi-automatic manual segmentation methods in identifying dominant and index tumors [62]. Integrating AI-driven tools, such as aPROMISE, into the PSMA PET workflow can improve standardized reporting, efficiency, and diagnostic accuracy. However, larger prospective studies are required to validate its performance and support clinical adoption [63]. AI is not a replacement but a valuable adjunct, particularly in settings with limited access to nuclear medicine specialists.
Furthermore, the AI space is rapidly evolving with advances being made beyond basic anatomical segmentation. PSMA-based AI has been able to assist with theranostic treatment response and differentiate between intraprostatic disease and more advanced grades including extra-prostatic extension. However, there are still a number of obstacles to broader adoption including a lack of standardized protocols, the need for further validation studies, and improved integration into clinical workflows [64,65].
Adoption of PSMA PET for Staging to Guide Clinical Decision-Making: Overview of the Unmet Need
Despite the growing evidence of PSMA PET for detecting prostate cancer lesions, its adoption is inconsistent across clinical practice [66]. An area that lacks robust data is the utilization of serial PSMA PET imaging in assessing patient response/progression and in guiding management [67]. The lack of validation studies has hindered broader adoption into routine clinical decision-making. In this context, some treating physicians have limited its use to patient selection and response assessment when using PSMA-directed targeted therapy. Recently, the Response Evaluation Criteria in PSMA PET/CT (RECIP 1.0) has been developed to standardize the assessment of systemic treatment response in metastatic prostate cancer using PSMA PET/CT [68]. In addition, the recently published Prostate Cancer Clinical Trials Working Group 4 (PCWG4) guidance recommends new criteria for the use of PSMA PET in validating treatment response and progression in prospective clinical trials [69,70]. Given the limited clinical evidence linking PSMA PET findings to improved therapeutic outcomes, some physicians may continue relying on conventional imaging.
Though PSMA PET is increasingly used for patient selection, conventional imaging remains the standard for follow-up response assessment in prostate cancer trials, despite its limitations in detecting lesions [67]. A key unmet need is the ability to determine high-and low-volume metastatic hormone-sensitive prostate cancer using PSMA PET, which might help identify patients who could benefit from the addition of docetaxel chemotherapy. In addition, it is not uncommon for patients selected based on PSMA-positive scans to lack corresponding target lesions on conventional imaging, complicating response assessment. As a result, some recent trials (such as ARROW, NCT03939689; [71]) have begun evaluating PSMA PET alongside bone and CT scans during therapy assessment. Future studies should directly compare conventional imaging and PSMA PET in a head-to-head design, generating essential conversion data that will facilitate the gradual transition from conventional imaging as more evidence accrues.
Addressing this unmet need
PSMA PET has significantly influenced the management of prostate cancer across different risk categories. Studies evaluating the clinical utility of PSMA PET have demonstrated changes in staging, treatment planning, and therapeutic strategy [72].
Unfavorable intermediate-risk disease
While most clinical trials have focused on high-risk or recurrent disease, recent evidence suggests that PSMA PET may also influence management decisions in patients with unfavorable intermediate-risk prostate cancer. In this group, PSMA PET has revealed nodal involvement or distant metastases that were not detected on conventional imaging, leading to changes in treatment [73]. A retrospective multicenter analysis reported a higher positivity rate for nodal and distant metastases in unfavorable intermediate-risk patients compared to favorable intermediate-risk patients, with PSMA PET impacting therapeutic management in 13.3% of cases overall [74]. The PSMA-dRT trial, a multicenter phase 3 study, evaluated PSMA PET before definitive radiotherapy in patients with unfavorable intermediate- and high-risk prostate cancer [75]. PSMA PET/CT upstaged 17% of patients relative to baseline staging, which resulted in more accurate radiotherapy planning, including radiation volume delineation, dose escalation, and hormone therapy intensification. Unfavorable intermediate-risk prostate cancer is at the crossroads of indolent and aggressive disease and can benefit significantly from the precise staging of PSMA PET and optimizing strategies.
Though PSMA PET is approved for staging in unfavorable intermediate-risk prostate cancer, it is not routinely recommended for favorable intermediate-risk disease [22]. However, emerging data suggest that PSMA PET may add value in detecting atypical metastases and false-negative lesions from CT or bone scan in this population [76]. These findings highlight a potential overlap between favorable and unfavorable intermediate-risk disease. Future studies are needed to clarify the utility of PSMA PET in favorable intermediate-risk prostate.
High-risk disease
In high-risk prostate cancer patients, PSMA PET has demonstrated higher sensitivity over conventional imaging in detecting metastatic disease in pelvic and distant lymph nodes [10,77]. Studies have shown that PSMA PET significantly impacts management decisions, leading to changes in radiation planning and systemic therapy initiation or escalation in high-risk prostate cancer patients [75,77,78].
The proPSMA trial, a multicenter randomized study, found that PSMA PET/CT altered management plan in 28% of high-risk patients, influencing strategies such as shifting from curative-intent surgery to systemic therapy [77].
Recently, a study evaluated the usefulness of the PSMA scores in reporting PSMA PET/CT in high-risk prostate cancer patients [79]. It reported a change in disease staging in 39% of cases following PSMA PET/CT, and PSMA score leading to a change in therapeutic decision-making in 32% of patients [79]. These findings underscore the crucial role of PSMA PET in optimizing treatment strategies and guiding clinical management of high-risk patients.
Biochemical recurrence
In patients with BCR following definitive therapy, PSMA PET has revealed previously undetected local recurrences [80], as well as nodal involvement and distant metastases [12,80,81], even at low PSA levels [81]. The identification of locoregional versus distant recurrence directly influences salvage strategies, guiding treatment tailored to the disease burden. The CONDOR study, which evaluated piflufolastat F 18 PET in patients with rising PSA and non-informative standard imaging, demonstrated a change in intended management in 63.9% of BCR cases following PSMA PET [81]. These changes included transitions from observation to initiating therapy, shifts between systemic and local therapies, and a shift from planned treatment to observation [81]. An Australian prospective multicenter study found that PSMA PET altered management intent in 62% of BCR patients, distinguishing locoregional recurrence in 39% of cases from distant metastases in 16% of cases [82]. In patients with very early BCR, PSMA PET led to management changes in 42.8% of cases, including salvage radiotherapy in select patients with pelvic oligometastases and initiation of systemic therapy in cases where extra-pelvic disease was detected [83]. These findings highlight the role of PSMA PET in accurately localizing recurrence, improving salvage therapy decisions, and preventing empiric treatment that lacks precise disease identification.
Despite the encouraging impact of PSMA PET on clinical decision-making, direct evidence linking its use to improved patient outcomes remains limited. One prospective study demonstrated that PSMA PET findings were highly predictive of 3-year freedom from progression in post-prostatectomy BCR patients undergoing salvage radiotherapy [84]. While several studies have shown association between PSMA PET findings and subsequent management changes across different risk categories, definitive improvements in overall survival, progression-free survival, or quality of life have not yet been confirmed in prospective randomized trials. Larger, multicenter, outcome-driven studies are needed to establish the clinical benefit of PSMA PET and guide its broader adoption. Table 2 summarizes selected ongoing clinical trials of PSMA PET-guided therapy in prostate cancer with patient outcome measures.
Selected Ongoing Trials of PSMA PET-Guided Therapy in Prostate Cancer with Patient Outcome Measures
| Trial Name | Clinical Trial ID, Phase & Sponsor | Study focus | Population | Primary Outcomes | Secondary Outcomes | Imaging Agent | Estimated Readout |
|---|---|---|---|---|---|---|---|
| PSMA-SRT | NCT03582774 Phase 3 Sponsor: Jonsson Comprehensive Cancer Center | PSMA PET/CT-guided salvage RT planning post-prostatectomy | Biochemical recurrence after primary prostatectomy (n=193) | 5-year Biochemical PFS | Metastasis-free survival, initiation of additional salvage therapy, change in initial treatment intent | 68Ga-PSMA-11 | Primary completion: July 2026 Study completion: July 2027 |
| PSMA-PETgRT | NCT03525288 Phase 2/3 Sponsor: Centre hospitalier de l'Université de Montréal (CHUM) | PSMA PET-guided intensification of salvage RT | Biochemical recurrence following radical prostatectomy (n=130) | 5-year Failure-free survival | Rate of failure, toxicity, survival, QoL, detection yield of PSMA PET | 18F-DCFPyL | Primary completion: May 2026 Study completion: May 2027 |
| MIDAS-Prostate | NCT05328505 Phase 2 Sponsor: University Health Network, Toronto | PSMA PET-guided dose escalation/de-escalation in salvage RT | Post-prostatectomy BCR with PSMA PET-confirmed locoregional recurrence (n=80) | Grade ≥2 Toxicity for GU | Biochemical failure-free survival, QoL | PSMA PET (unspecified) | Primary completion: August 2028 Study completion: August 2030 |
| EMPIRE-2 | NCT03762759 Phase 2 Sponsor: Emory University | Dose escalation using PSMA PET vs 18F-fluciclovine PET post-prostatectomy | PSA recurrence following radical prostatectomy with negative conventional imaging (n=140) | 2-year Disease-free survival | Decision to offer radiotherapy, decision to treat pelvic nodes, decision to boost between the initial and final treatment decisions, prostate bed clinical target volume (CTV) and planning target volume (PTV) | 68Ga-PSMA vs. 18F-fluciclovine | Primary completion: May 2025 Study completion: December 2025 |
| PSMA recidiv | NCT04794777 Phase 3 Sponsor: Stefan Carlsson | Individualized PSMA PET-guided therapy vs standard salvage RT | BCR post-prostatectomy (n=450) | Primary PSA progression free survival | Time to metastasis, Prostate cancer specific survival, Time to secondary treatment, Differences in quality of life recorded using Patient Reported Outcome Measure (PROM) | 68Ga-PSMA-11 or 18F-PSMA-1007 | Primary completion: October 2027 Study completion: October 2027 |
| ARASTEP | NCT05794906 Phase 3 Sponsor: Bayer | PSMA PET-guided systemic therapy (darolutamide + ADT) in high-risk biochemical recurrence | High-risk BCR with PSMA PET-positive lesions, no evidence of metastases on conventional imaging (n=970) | Radiologic PFS by PSMA PET/CT | Metastasis-free survival, time to CRPC, time to SSE, overall survival, QoL | PSMA PET-CT (tracer not specified) | Primary completion: July 2027 Study completion: March 2030 |
Patient selection and post-treatment assessment
As it relates to patient selection and post-treatment assessment for PSMA-targeted radioligand therapy (RLT), critically important guidelines have just been published by the Prostate Cancer Working Group 4 (PCWG4) [70]. Specifically, while provided within the context of prostate cancer clinical trials, the guidance recommends baseline PSMA-PET/CT for pretreatment imaging whenever feasible. This is essential information given the additional recommendation to expand response and progression criteria as a proposed method to test and validate in the clinical trial setting. Observed changes in SUVmax, SUVmean, or SUV-assessed tumor volume will help determine the extent of treatment response not just to RLT but potentially, non-PSMA targeted treatment paradigms.
As well as PCWG4, Gafita et al., have described the use of SUVmax, SUVmean, or SUV-assessed tumor volume for treatment response evaluation [67]. PERCIST, first developed for FDG PET/CT using SUVpeak, has been modified for PSMA PET with mixed results. In addition, PSMA PET progression (PPP) and RECIP 1.0 criteria have been developed to leverage these measurements to evaluate treatment response. PPP criteria include the appearance of 2 or more new distant lesions that are PSMA-positive or a new lesion consistent with clinical or lab findings and histopathology confirmation or corroborative imaging. Alternatively, RECIP 1.0 defines a new lesion as new focal uptake of a PSMA-targeted agent that is higher than surrounding background with a tumor lesion SUVmax greater than blood-pool SUVmax; its accompanying classification system allows for assessments of tumor responses from progressive disease to complete response [67].
To address the challenge of variability in SUV measurements across different scanners, tracers, and protocols, ongoing efforts, such as EANM/European Association of Nuclear Medicine Research Ltd. (EARL) accreditation, and programs by EARL and the SNMMI Clinical Trials Network (CTN), are aimed at making these metrics robust enough for multi-center trials. Quality assurance and control focused on daily calibration measurements and cross-calibration for F 18- and Ga 68-based agents have been previously described as well as PET/CT manufacturer-based guidance [23,85].
Lastly, it will be interesting to see how these emerging PSMA-based criteria will compare with conventional imaging (e.g., RECIST) and other functional modalities such as FDG PET in the context of future clinical studies. In a recent head-to-head comparison of 18F-FDG and PSMA PET/CT, both sensitivity and diagnostic accuracy were greater for PSMA at both 1 and 3 hour imaging time points [86].
Potential role of PSMA PET-derived metrics in staging and clinical decision-making
SUVmax, in particular, has emerged as a useful biomarker in prostate cancer imaging and plays an important role in tumor characterization, risk stratification, and treatment decision-making. Several studies have demonstrated a positive correlation between SUVmax and clinical parameters such as Gleason score, PSA levels, and tumor aggressiveness [87,88]. Higher SUVmax values observed in the prostate gland and metastatic lesions are often associated with more aggressive disease and higher-grade tumors.
In diagnostic contexts, SUVmax has demonstrated utility in differentiating malignant from benign lesions, guiding targeted biopsies, and selecting candidates for focal therapies or systemic treatment escalation [89,90]. A recent study retrospectively analyzed thirty-four 68Ga-PSMA-11 PET/CT studies performed during the initial staging and reported a positive correlation between intraprostatic SUVmax and the WHO/ISUP grade group of prostate cancer [91]. The study revealed that high-risk patients exhibited significantly higher mean SUVmax values compared with low-risk patients, reinforcing the role of SUVmax in risk stratification.
SUVmax may also help identify suspicious lesions warranting targeted biopsy. A pre-biopsy PSMA PET/CT scan may guide the biopsy site, potentially reducing false negative results [89]. Furthermore, detection of lesions with markedly elevated SUVmax may lead to intensified treatment strategies such as surgery or systemic therapy, while low-uptake foci in the prostate support active surveillance.
Beyond staging, SUVmax has been associated with the pathologic upgrading from non-clinically significant to clinically significant prostate cancer between biopsy and surgery [92]. A prospective real-world study established and validated the optimal SUVmax cutoff value of 5.3 for distinguishing clinically significant prostate cancer from benign prostate disease [93].
Moreover, SUVmax is gaining attention for its ability to predict treatment response and clinical prognosis. Recent evidence suggests that baseline SUVmax is closely associated with biochemical response and may help in identifying responders to PSMA-targeted radioligand therapy [94]. Overall, these findings emphasize the expanding role of SUVmax in the personalized management of prostate cancer.
In addition to SUVmax, other PSMA PET-derived metrics, such as whole-body SUVmean and total tumor volume (TTV), are recognized as important emerging biomarkers [48,95]. Baseline whole-body SUVmean has been shown to predict treatment response to 177Lu-PSMA-617 therapy in mCRPC [48,49,96], while PSMA-TTV has demonstrated prognostic value for clinical outcomes by providing a comprehensive measure of disease burden [47,94]. Together, these PSMA PET metrics can complement each other in guiding treatment decisions.
Expert Perspectives
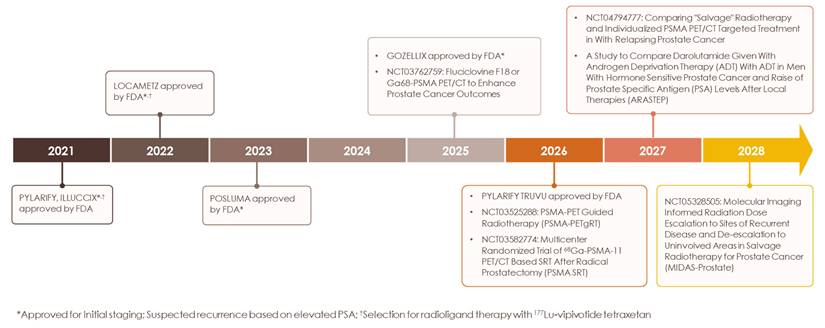
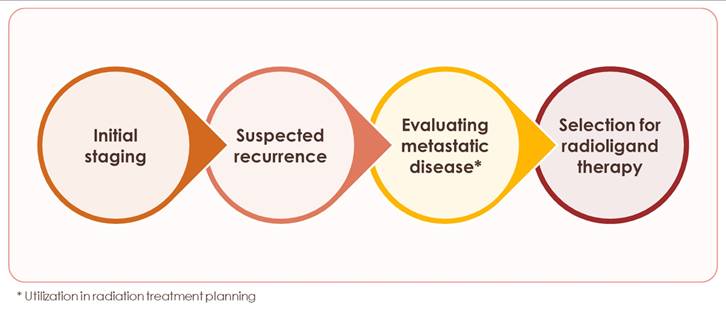
Starting with the initial FDA approval of Ga 68 PSMA-11 for UCLA/UCSF in December 2020 [97,98], PSMA PET has transformed prostate cancer imaging (Figure 3) and, in the process, patient management. We have entered a new era of prostate cancer diagnostics marked by higher accuracy and improved detection of previously unidentified lesions compared to conventional modalities. The recommended applications of PSMA PET continue to expand and include: initial staging, detecting biochemical recurrence, and evaluating metastatic disease (Figure 4) [23]. For patients with high-risk prostate cancer, some centers now proceed directly to PSMA PET, bypassing conventional bone or CT scans, a practice supported by updated guidelines. Although PSMA PET is not routinely recommended for low-risk localized prostate cancer, it may be valuable in certain scenarios, such as clarifying inconclusive MRI findings or investigating an unexplained PSA elevation despite negative conventional imaging or biopsy results. This nuanced, case-by-case use underscores the importance of clinical judgment and highlights the need for continued education on evidence-based imaging to avoid both underuse and overuse.
Timeline of PSMA PET agent milestones. History of FDA approval for current PSMA PET radiotracers and ongoing clinical trials evaluating current and novel radiotracers in diverse indications.
Applications of PSMA PET for prostate cancer. Applications where use of PSMA PET is currently recommended.
In standard practice, PSMA PET is often used for patient selection and eligibility assessment; however, treatment response is still typically evaluated using conventional imaging, even when these modalities initially fail to detect disease. In clinical trial design, we strongly encourage the use of the same imaging agent throughout the study, from baseline to follow-up, to ensure consistency and enable optimal interpretation.
Different tracers exhibit distinct uptake and distribution patterns; therefore, the choice of imaging agent can significantly influence SUV interpretation. This variability may complicate lesion characterization, particularly in oligometastatic disease. Despite the established role of PSMA PET in prostate cancer, further prospective data are needed to define its utility in intermediate-risk and oligometastatic settings [99,100].
With the publication of PCWG4 [70], a consensus framework for incorporating PSMA PET with clinical and PSA criteria, new guidance on its use for treatment response and its integration into clinical trials will help facilitate the integration of meaningful parameters in this critically needed space. These insights will help to provide the necessary context for this cutting-edge guidance.
We also would like to acknowledge advancements in non-imaging biomarker methods. Liquid biopsies are minimally invasive and can offer a complementary, comprehensive systemic approach. The use of serum and urinary biomarkers, such as circulating tumor cells and DNA fragments, offer valuable insights into the molecular composition of tumors. Within this context, the extraction and analysis of exosomes from liquid biopsy samples, such as blood, urine, and semen, have demonstrated significant potential as a source of novel biomarkers for prostate cancer [101].
Lastly, another domain that has gained increasing attention in recent years is the role of PSMA PET in non-prostate malignancies. PSMA is also expressed on endothelial cells within the neovasculature of other solid tumors [102]. Emerging evidence demonstrates promising results with PSMA PET in renal cell carcinoma [103], breast cancer [104], glioblastoma [105], and hepatocellular carcinoma [106], suggesting potential avenues for its diagnostic and therapeutic use in non-prostate cancers. This also brings up the importance of addressing PSMA-negative or low-expressing lesions in prostate cancer itself and the need for a future multi-targeted approach that could include promising non-PSMA biomarkers such as cluster of differentiation 46 (CD46), delta-like ligand 3 (DLL3), fibroblast activation protein (FAP), gastrin releasing peptide receptor (GRPR), and human kallikrein 2 (hK2) [107].
Conclusion
PSMA PET has revolutionized the diagnosis and management of prostate cancer, with its role established in some settings while still evolving in others. Barriers to broader adoption across different clinical settings and among practitioners can be addressed through continued education and research. In the future, integration of AI-based tools or algorithms into routine PSMA PET workflow may enhance efficiency, reproducibility, diagnostic accuracy, and personalized treatment approaches.
Abbreviations
MD PET: multi-disciplinary PSMA expert team; PSMA: prostate-specific membrane antigen; CT: computed tomography; MRI: magnetic resonance imaging; PET: positron emission tomography; FDA: food and drug administration; NCCN: national comprehensive cancer network; VA: veteran affairs; SNMMI: society of nuclear medicine and molecular imaging; mCRPC: metastatic castration-resistant prostate cancer; SUV: standardized uptake value; BCR: biochemical recurrence; AI: artificial intelligence; RECIP: response evaluation criteria in PSMA PET/CT; PCWG4: prostate cancer clinical trials working group 4; TTV: total tumor volume.
Acknowledgements
This manuscript was endorsed by ACRO on April 7, 2026. No AI tools were used for the writing of this manuscript.
Disclosures
Lantheus provided medical writing support and article processing charges on behalf of all authors. Technical support for the graphical abstract and manuscript figures was funded by Lantheus and provided by Anastasia Hains, PhD, of Nucleus Global, an Inizio company. C.M.P. reports stock and other ownership Interests: US Urology Partners; ImmunityBio; Honoraria: Janssen; Dendreon; Pfizer/Astellas; Bayer; Sun Pharma; Myovant Sciences; Merck; AstraZeneca; Bristol-Myers Squibb; Novartis; ImmunityBio; consulting or advisory Role: Pfizer/Astellas; Bayer; Janssen Oncology; Tolmar; Sun Pharma; Dendreon; Merck; AstraZeneca; Bristol-Myers Squibb; Novartis; ImmunityBio; speakers' bureau: Bayer; Dendreon; Pfizer; Astellas Pharma; Sun Pharma; Myovant Sciences; Janssen Oncology; Merck; Research Funding: Bayer, Pfizer; Astellas Pharma; Merck; AstraZeneca; Advantagene; Dendreon; Janssen Oncology; InVitae; Candel Therapeutics (institution); CG Oncology (institution); ImmunityBio (institution). E.Y.Y. reports consulting roles for Astellas, Johnson & Johnson, AstraZeneca, Tolmar 2, Merck, Bayer, Lantheus, Novartis, Bristol-Myers Squibb, Loxo; and research funding to institution (University of Washington) from Dendreon, Merck, SeaGen/Pfizer, Blue Earth, Bayer, Lantheus, and Tyra. S.E.F. reports stock and other ownership interests US Urology Partners; NWDH I, NWDH II; 2100 State Acquisition, Jellyfish Health, ImmunityBio; Histosonics; consulting & honoraria: Astellas, Bayer, Blue Earth, ImmunityBio, Jansen, Lantheus, Pfizer. Progenics; Institutional Research Funding thru role with.US Urology Partners with Bayer, Pfizer; Astellas Pharma; Merck; AstraZeneca; Advantagene; Dendreon; Janssen Oncology; InVitae; Candel Therapeutics; CG Oncology ; ImmunityBio; Novartis; leadership role: ACRO (American College of Radiation Oncology).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-63
2. Siegel RL, Kratzer TB, Wagle NS, Sung H, Jemal A. Cancer statistics, 2026. CA Cancer J Clin. 2026;76:e70043
3. National Cancer Institute. Cancer Stat Facts: Prostate Cancer. Updated 2023. https://seer.cancer.gov/statfacts/html/prost.html
4. Attard G, Murphy L, Clarke NW, Sachdeva A, Jones C, Hoyle A. et al. Abiraterone acetate plus prednisolone with or without enzalutamide for patients with metastatic prostate cancer starting androgen deprivation therapy: final results from two randomised phase 3 trials of the STAMPEDE platform protocol. Lancet Oncol. 2023;24:443-56
5. Tapper W, Carneiro G, Mikropoulos C, Thomas SA, Evans PM, Boussios S. The application of radiomics and AI to molecular imaging for prostate cancer. J Pers Med. 2024;14:287
6. Hövels AM, Heesakkers RA, Adang EM, Jager GJ, Strum S, Hoogeveen YL. et al. The diagnostic accuracy of CT and MRI in the staging of pelvic lymph nodes in patients with prostate cancer: a meta-analysis. Clin Radiol. 2008;63:387-95
7. Tanaka T, Yang M, Froemming AT, Bryce AH, Inai R, Kanazawa S. et al. Current imaging techniques for and imaging spectrum of prostate cancer recurrence and metastasis: a pictorial review. Radiographics. 2020;40:709-726
8. Hope TA, Benz M, Jiang F, Thompson D, Barbato F, Juarez R. et al. Do bone scans overstage disease compared with PSMA PET at initial staging? An international multicenter retrospective study with masked independent readers. J Nucl Med. 2023;64:1744-1747
9. Hope TA, Eiber M, Armstrong WR, Juarez R, Murthy V, Lawhn-Heath C. et al. Diagnostic accuracy of 68Ga-PSMA-11 PET for pelvic nodal metastasis detection prior to radical prostatectomy and pelvic lymph node dissection: a multicenter prospective phase 3 imaging trial. JAMA Oncol. 2021;7:1635-1642
10. Pienta KJ, Gorin MA, Rowe SP, Carroll PR, Pouliot F, Probst S. et al. A phase 2/3 prospective multicenter study of the diagnostic accuracy of prostate specific membrane antigen PET/CT with 18F-DCFPyL in prostate cancer patients (OSPREY). J Urol. 2021;206:52-61
11. Hofman MS, Hicks RJ, Maurer T, Eiber M. Prostate-specific membrane antigen PET: clinical utility in prostate cancer, normal patterns, pearls, and pitfalls. Radiographics. 2018;38:200-217
12. Holzgreve A, Armstrong WR, Clark KJ, Benz MR, Smith CP, Djaileb L. et al. PSMA-PET/CT findings in patients with high-risk biochemically recurrent prostate cancer with no metastatic disease by conventional imaging. JAMA Netw Open. 2025;8(1):e2452971
13. Ghosh A, Wang X, Klein E, Heston WD. Novel role of prostate-specific membrane antigen in suppressing prostate cancer invasiveness. Cancer Res. 2005;65:727-31
14. Maes J, Gesquière S, De Spiegeleer A, Maes A, Van de Wiele C. Prostate-specific membrane antigen biology and pathophysiology in prostate carcinoma, an update: potential implications for targeted imaging and therapy. Int J Mol Sci. 2024;25:9755
15. PYLARIFY® (piflufolastat F 18) prescribing information. Progenics Pharmaceuticals, Inc, a Lantheus company. Updated 2021. https://pylarify.com/sites/default/files/resources/prescribing-information.pdf
16. PYLARIFY TRUVUTM (piflufolastat F 18) prescribing information. Aphelion LLC. 2026. https://pylarifytruvuhcp.com/.
17. POSLUMA® (flotufolastat F 18) prescribing information. Blue Earth Diagnostics Ltd. Updated 2024. https://www.posluma.com/prescribing-information.pdf
18. ILLUCCIX® (kit for the preparation of gallium Ga 68 gozetotide injection) prescribing information. Telix Pharmaceuticals (US), Inc. Updated 2025. https://illuccix.com/wp-content/uploads/2025/03/Illuccix-PI-9.11.2025.pdf
19. LOCAMETZ® (kit for the preparation of gallium Ga 68 gozetotide injection) prescribing information. Advanced Accelerator Applications USA, Inc. Updated 2025. https://www.novartis.com/us-en/sites/novartis_us/files/locametz.pdf
20. GOZELLIXTM (kit for the preparation of gallium Ga 68 gozetotide injection) prescribing information. Telix Pharmaceuticals (US), Inc. Updated 2025. https://telixpharma.com/wp-content/uploads/2025/03/Gozellix-prescribing-information.pdf
21. PLUVICTO® (lutetium Lu 177 vipivotide tetraxetan) prescribing information. Novartis Pharmaceuticals Corporation. Updated 2025. https://www.novartis.com/us-en/sites/novartis_us/files/pluvicto.pdf
22. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Prostate Cancer. Version 5.2026. https://www.nccn.org/guidelines/guidelines-detail?id=1459
23. Fendler WP, Eiber M, Beheshti M, Bomanji J, Calais J, Ceci F. et al. PSMA PET/CT: joint EANM procedure guideline/SNMMI procedure standard for prostate cancer imaging 2.0. Eur J Nucl Med Mol Imaging. 2023;50:1466-1486
24. Crawford ED, Albala DM, Harris RG, Slovin SF, Bryce AH, Carroll PR. et al. A clinician's guide to targeted precision imaging in patients with prostate cancer (RADAR VI). JU Open Plus. 2023;1(1):e00003
25. Keppler JS, Conti PS. A cost analysis of positron emission tomography. AJR Am J Roentgenol. 2001;177:31-40
26. Miller SR, Gonzalez RT, Jackson WC, Caram MEV, Tsao PA, Stensland K. et al. Rates of PSMA PET staging and positivity in newly diagnosed prostate cancer in a national health care system. J Nucl Med. 2025;66:75-83
27. Czernin J, Adams T, Calais J. More unacceptable denials: now it's PSMA-targeted PET/CT imaging. J Nucl Med. 2022;63:969-970
28. Borkowetz A, Linxweiler J, Fussek S, Wullich B, Saar M; German prostate cancer consortium (DPKK). The role of PSMA PET imaging in prostate cancer theranostics: a nationwide survey. Urol Int. 2022;106:1126-1135
29. Chiong E, Saad M, Hamid ARAH, Ong-Cornel AB, Lojanapiwat B, Pripatnanont C. et al. Prostate cancer management in Southeast Asian countries: a survey of clinical practice patterns. Ther Adv Med Oncol. 2024;16:1-20
30. Schenzle C. Barriers and solutions in cross-sector care for metastatic prostate cancer patients in Germany: a qualitative study on radioligand therapy. BMC Health Serv. Res. 2025;25:1281
31. Hussain D, Abbas N, Khan J. Recent breakthroughs in PET-CT multimodality imaging: innovations and clinical impact. Bioengineering (Basel). 2024;11:1213
32. Abass LA, Usuemerai PA, Ibikunle OE, Alemede V, Mbata AO. Public-private partnership to enhance healthcare access and affordability. Int J Multidiscip Res Growth Eval. 2024;5:1327-1344 doi:10.54660/.IJMRGE.2024.5.4.1327-1344
33. National Cancer Institute. NCI-Designated Cancer Centers. Updated March 19, 2025. https://www.cancer.gov/research/infrastructure/cancer-centers
34. Privé BM, Govers TM, Israël B, Janssen MJR, Timmermans BJR, Peters SMB. et al. A cost-effectiveness study of PSMA-PET/CT for the detection of clinically significant prostate cancer. Eur J Nucl Med Mol Imaging. 2025;52:3159-3169
35. Yee CW, Harvey MJ, Xin Y. Kirson NY. Cost-effectiveness modeling of prostate-specific membrane antigen positron emission tomography with piflufolastat F 18 for the initial diagnosis of patients with prostate cancer in the United States. Pharmacoeconomics. 2024;42:231-247
36. Hoffman A, Amiel GE. The impact of PSMA PET/CT on modern prostate cancer management and decision making-the urological perspective. Cancers (Basel). 2023;15:3402
37. Islam R, Desai S, Moran M, Golombos DM. The role of PSMA PET imaging in prostate cancer: current applications and future directions. Curr Urol Rep. 2025;26:46
38. Sunderland JJ, Christian PE. Quantitative PET/CT scanner performance characterization based upon the society of nuclear medicine and molecular imaging clinical trials network oncology clinical simulator phantom. J Nucl Med. 2015;56:145-52
39. Martiniova L, Zurita A, Pan T, Ravizzni G, Mawlawi O. Standardizing PSMA-PET-based response assessment across different PET/CT imaging platforms. J Nucl Med. 2025;66(Suppl 1):252185
40. Alberts I, Prenosil G, Sachpekidis C, Weitzel T, Shi K, Rominger A, Afshar-Oromieh A. Digital versus analogue PET in [68Ga]Ga-PSMA-11 PET/CT for recurrent prostate cancer: a matched-pair comparison. Eur J Nucl Med Mol Imaging. 2020;47:614-623
41. Rovera G, Grimaldi S, Dall'Armellina S, Zotta M, Finessi M, Passera R. et al. Comparison of digital versus analog 68Ga-PSMA-11 PET/CT performance in hormone-sensitive prostate cancer patients with early biochemical recurrence or persistence after radical treatment. Diagnostics (Basel). 2023;13:3535
42. Donswijk ML, Ettema RH, van der Gaag S, Wondergem M, Cheung Z, van der Poel HG. et al. The intra- and interobserver variability of PSMA-expression scores in patients with primary prostate cancer. EJNMMI Res. 2024;14:99
43. Donswijk ML, Ettema RH, Meijer D, Wondergem M, Cheung Z, Bekers EM. et al. The accuracy and intra- and interobserver variability of PSMA PET/CT for the local staging of primary prostate cancer. Eur J Nucl Med Mol Imaging. 2024;51:1741-1752
44. Yazdanpanah F, Mhana SAA, Werner T, Alavi A, Revheim ME. Comparison of [18F]PSMA-1007, [18F]DCFPyL, and [68Ga]Ga-PSMA-11 in PET/CT imaging for prostate cancer: a systematic review and meta-analysis. J Nucl Med. 2025;66(Suppl 1):251575
45. Donswijk ML, Wondergem M, de Wit-van der Veen L, Bruin NM, van Leeuwen PJ, van der Poel HG. et al. Effects of furosemide and tracer selection on urinary activity and peri-bladder artefacts in PSMA PET/CT: a single-centre retrospective study. EJNMMI Res. 2022;12(1):42
46. Murthy V, Smith RL, Tao DH, Lawhn-Heath CA, Korenchan DE, Larson PEZ. et al. 68Ga-PSMA-11 PET/MRI: determining ideal acquisition times to reduce noise and increase image quality. EJNMMI Phys. 2020;7:54
47. Esfahani SA, Morris MJ, Sartor O, Frydenberg M, Fanti S, Calais J. et al. Standardized template for clinical reporting of PSMA PET/CT scans. Eur J Nucl Med Mol Imaging. 2024;52:335-341
48. Kuo PH, Morris MJ, Hesterman J, Kendi AT, Rahbar K, Wei XX. et al. Quantitative 68Ga-PSMA-11 PET and clinical outcomes in metastatic castration-resistant prostate cancer following 177Lu-PSMA-617 (VISION Trial). Radiology. 2024;312:e233460
49. Buteau JP, Martin AJ, Emmett L, Iravani A, Sandhu S, Joshua AM. et al. PSMA and FDG-PET as predictive and prognostic biomarkers in patients given [177Lu]Lu-PSMA-617 versus cabazitaxel for metastatic castration-resistant prostate cancer (TheraP): a biomarker analysis from a randomised, open-label, phase 2 trial. Lancet Oncol. 2022;23:1389-1397
50. Graham MM. The future of nuclear medicine in the United States. J Nucl Med. 2023;64:1352-1353
51. Nensa F, Demircioglu A, Rischpler C. Artificial intelligence in nuclear medicine. J Nucl Med. 2019;60(Suppl 2):29S-37S
52. Shaygan B, Zukotynski K, Bénard F, Ménard C, Kuk J, Sistani G. et al. Canadian Urological Association best practice report: prostate-specific membrane antigen positron emission tomography/computed tomography (PSMA PET/CT) and PET/magnetic resonance (MR) in prostate cancer. Can Urol Assoc J. 2021; 15: 162-172. Erratum in: Can Urol Assoc J. 2021;15:E423
53. Heilinger J, Roth KS, Weis H, Fink A, Weindler J, Dietlein F. et al. Do you know your PSMA-tracer? Variability in the biodistribution of different PSMA ligands and its potential impact on defining PSMA-positivity prior to PSMA-targeted therapy. EJNMMI Res. 2025;15:4
54. Piron S, De Man K, Schelfhout V, Van Laeken N, Kersemans K, Achten E. et al. Optimization of PET protocol and interrater reliability of 18F-PSMA-11 imaging of prostate cancer. EJNMMI Res. 2020;10:14
55. Hohberg M, Kobe C, Täger P, Hammes J, Schmidt M, Dietlein F. et al. Combined early and late [68Ga]PSMA-HBED-CC PET scans improve lesion detectability in biochemical recurrence of prostate cancer with low PSA levels. Mol Imaging Biol. 2019;21:558-566
56. Derlin T, Weiberg D, von Klot C, Wester HJ, Henkenberens C, Ross TL. et al. 68Ga-PSMA I&T PET/CT for assessment of prostate cancer: evaluation of image quality after forced diuresis and delayed imaging. Eur Radiol. 2016;26:4345-4353
57. Rauscher I, Langbein T, Fuetterer C, Weber W, Eiber M. Evaluation of qualitative and quantitative PET parameters in primary prostate cancer patients: double-match comparison of 18F-flotufolastat-and 68Ga-PSMA-PET. J Nucl Med. 2024;65(Suppl 2):241424
58. Voter AF, Werner RA, Savas H, Gafita A, Ross AE, Gorin MA. et al. A practical guide to the pearls and pitfalls of PSMA PET imaging. Semin Nucl Med. 2024;54:119-131
59. Emmett L, Zheng A, Jin C, Papa N, Khan S, Ayati N. et al. 'One button push' fully automated PSMA PET quantification: correlation with progression-free and overall survival in patients undergoing [177Lu] Lu PSMA therapy for metastatic castration-resistant prostate cancer. J Clin Oncol. 2025;43(Suppl 16):5054
60. Nickols N, Anand A, Johnsson K, Brynolfsson J, Borreli P, Parikh N. et al. aPROMISE: a novel automated PROMISE platform to standardize evaluation of tumor burden in 18F-DCFPyL images of veterans with prostate cancer. J Nucl Med. 2022;63:233-239
61. Johnsson K, Brynolfsson J, Sahlstedt H, Nickols NG, Rettig M, Probst S. et al. Analytical performance of aPROMISE: automated anatomic contextualization, detection, and quantification of [18F]DCFPyL (PSMA) imaging for standardized reporting. Eur J Nucl Med Mol Imaging. 2022;49:1041-1051
62. García Vicente AM, Lucas Lucas C, Pérez-Beteta J, Borrelli P, García Zoghby L, Amo-Salas M. et al. Analytical performance validation of aPROMISE platform for prostate tumor burden, index and dominant tumor assessment with 18F-DCFPyL PET/CT. A pilot study. Sci Rep. 2024;14:3001
63. Dadgar H, Hong X, Karimzadeh R, Ibragimov B, Majidpour J, Arabi H. et al. The value of artificial intelligence in PSMA PET: a pathway to improved efficiency and results. Q J Nucl Med Mol Imaging. 2025;69:157-173
64. Liu J, Sandhu K, Woon DTS, Perera M, Lawrentschuk N. The value of artificial intelligence in prostate-specific membrane antigen positron emission tomography: an update. Semin Nucl Med. 2025;55:371-376
65. Usmani S, Al Riyami K, Kheruka S, Numani SP, Al Sukaiti R, Ahmed M. Deep learning (DL)-based advancements in prostate cancer imaging: artificial intelligence (AI)-based segmentation of 68Ga-PSMSA PET for tumor volume assessment. Precis Radiat Oncol. 2025;9:120-132
66. McKone EL, Sutton EA, Johnson GB, Phillips RM. Application of advanced imaging to prostate cancer diagnosis and management: a narrative review of current practice and unanswered questions. J Clin Med. 2024;13:446
67. Gafita A, Schroeder JA, Ceci F, Oldan JD, Khandani AH, Lecouvet FE. et al. Treatment response evaluation in prostate cancer using PSMA PET/CT. J Nucl Med. 2025;66:995-1004
68. Gafita A, Djaileb L, Rauscher I, Fendler WP, Hadaschik B, Rowe SP. et al. Response evaluation criteria in PSMA PET/CT (RECIP 1.0) in metastatic castration-resistant prostate cancer. Radiology. 2023;308:e222148
69. Hofman MS, Gafita A, Bressel M, Alipour R, Levy S, Emmett L. et al. Prostate cancer working group (PCWG4) preliminary criteria using serial PSMA PET/CT for response evaluation: analysis from the PRINCE trial. Ann Oncol. 2024;35(Suppl 2):S970
70. Armstrong AJ, Morris MJ, Abida W, Aggarwal RR, Antonarakis ES, Attard G. et al. Trial design and objectives for patients with prostate cancer: recommendations from the prostate cancer working group 4. J Clin Oncol. 2026Feb;26:JCO2502834 doi: 10.1200/JCO-25-02834. Online ahead of print
71. Yu EY, Narayan V, Esposito G, Szmulewitz R, Lu Y, Lilly MB. et al. 131I-LNTH-1095 radioligand therapy plus enzalutamide vs. enzalutamide alone in men with PSMA-avid metastatic castration-resistant prostate cancer: a phase 2 study. 2026 Mar 4. doi: 10.1158/1078-0432.CCR-25-4948. Epub ahead of print
72. Wang L, Wang L, Wang X, Wu D. The evolving role of PSMA-PET/CT in prostate cancer management: an umbrella review of diagnostic restaging, therapeutic redirection, and survival impact. Curr Oncol Rep. 2025;27:774-787
73. Basso Dias A, Finelli A, Bauman G, Veit-Haibach P, Berlin A, Ortega C. et al. Impact of 18F-DCFPyL PET on staging and treatment of unfavorable intermediate or high-risk prostate cancer. Radiology. 2022;304:600-608
74. Evangelista L, Guglielmo P, Giacoppo G, Setti L, Aricò D, Muraglia L. et al. The evaluation of radiolabeled prostate-specific membrane antigen positron emission tomography/computed tomography for initial staging in intermediate-risk prostate cancer patients: a retrospective multicenter analysis. Diagnostics (Basel). 2024;14:2751
75. Nikitas J, Lam E, Booker KA, Fendler WP, Eiber M, Hadaschik B. et al. Randomized trial of prostate-specific membrane antigen PET/CT before definitive radiotherapy for unfavorable intermediate- and high-risk prostate cancer (PSMA-dRT Trial). J Nucl Med. 2024;65:1076-1079
76. Green A, Temsah P, Alsagheer A, Abdalla A, Gu L, Muzaffar R. et al. 18F-PSMA PET/CT is helpful in the evaluation of intermediate risk favorable and low risk prostate cancer patients: A pictorial essay. J Nucl Med. 2023;64(Suppl 1):P1336
77. Hofman MS, Lawrentschuk N, Francis RJ, Tang C, Vela I, Thomas P. et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): a prospective, randomised, multicentre study. Lancet. 2020;395(10231):1208-1216
78. Ménard C, Young S, Zukotynski K, Hamilton RJ, Bénard F, Yip S. et al. PSMA PET/CT guided intensification of therapy in patients at risk of advanced prostate cancer (PATRON): a pragmatic phase III randomized controlled trial. BMC Cancer. 2022;22(1):251
79. Yasmin T, Younis MN, Ameer K, Farooq A, Shahid A. 68Ga-PSMA PET-CT and PSMA score affecting therapeutic decision-making in high-risk prostatic carcinoma. Pol J Radiol. 2023;88:e331-e337
80. Harsini S, Wilson D, Saprunoff H, Allan H, Gleave M, Goldenberg L. et al. Outcome of patients with biochemical recurrence of prostate cancer after PSMA PET/CT-directed radiotherapy or surgery without systemic therapy. Cancer Imaging. 2023;23(1):27
81. Morris MJ, Rowe SP, Gorin MA, Saperstein L, Pouliot F, Josephson D. et al. Diagnostic performance of 18F-DCFPyL-PET/CT in men with biochemically recurrent prostate cancer: results from the CONDOR phase III, multicenter study. Clin Cancer Res. 2021;27:3674-3682
82. Roach PJ, Francis R, Emmett L, Hsiao E, Kneebone A, Hruby G. et al. The impact of 68Ga-PSMA PET/CT on management intent in prostate cancer: results of an Australian prospective multicenter study. J Nucl Med. 2018;59:82-88
83. Bashir U, Tree A, Mayer E, Levine D, Parker C, Dearnaley D. et al. Impact of Ga-68-PSMA PET/CT on management in prostate cancer patients with very early biochemical recurrence after radical prostatectomy. Eur J Nucl Med Mol Imaging. 2019;46:901-907
84. Emmett L, Tang R, Nandurkar R, Hruby G, Roach P, Watts JA. et al. 3-year freedom from progression after 68Ga-PSMA PET/CT-triaged management in men with biochemical recurrence after radical prostatectomy: results of a prospective multicenter trial. J Nucl Med. 2020;61:866-872
85. Sunderland JJ, Boellaard R, Dickson JC, Graves SA, Bailey DL. Standardization of PET/CT performance requirements for whole-body quantitative imaging: an international proposal. J Nucl Med. 2025;66:1506-1515
86. Hu B, Yu H, Pan M, Yang H, Xing X, Li D. et al. Head-to-head comparison of 68Ga-PSMA-11 and 18F-FDG in delayed PET/CT imaging in prostate cancer diagnosis. Front Oncol. 2025;15:1515653
87. Uprimny C, Kroiss AS, Decristoforo C, Fritz J, von Guggenberg E, Kendler D. et al. 68Ga-PSMA-11 PET/CT in primary staging of prostate cancer: PSA and Gleason score predict the intensity of tracer accumulation in the primary tumour. Eur J Nucl Med Mol Imaging. 2017;44:941-949
88. Erdogan EB, Tekce E, Koca S, Aslan N, Toluk O, Aydin M. Predictive significance of intraprostatic volumetric parameters derived from early and standard time 68Ga-PSMA PET/CT images in newly diagnosed prostate cancer patients. Nucl Med Commun. 2024;45:629-641
89. Rahbar K, Weckesser M, Huss S, Semjonow A, Breyholz HJ, Schrader AJ. et al. Correlation of intraprostatic tumor extent with ⁶⁸Ga-PSMA distribution in patients with prostate cancer. J Nucl Med. 2016;57:563-567
90. Demirci E, Kabasakal L, Şahin OE, Akgün E, Gültekin MH, Doğanca T. et al. Can SUVmax values of Ga-68-PSMA PET/CT scan predict the clinically significant prostate cancer? Nucl Med Commun. 2019;40:86-91
91. Rogic I, Golubic AT, Zuvic M, Smitran T, Jukic N, Gamulin M. et al. Clinical utility of [68Ga]Ga-PSMA-11 PET/CT in initial staging of patients with prostate cancer and importance of intraprostatic SUVmax values. Nucl Med Rev Cent East Eur. 2024;27:6-12
92. Yang J, Tang Y, Zhu Z, Zhu H, Xiao L, Xiang S. et al. 68Ga-PSMA PET/CT SUVmax and its derivative in predicting clinical significance of prostate cancer or prostate cancer. J Nucl Med. 2022;63(Suppl 2):3067
93. Jiao J, Kang F, Zhang J, Quan Z, Wen W, Zhao X. et al. Establishment and prospective validation of an SUVmax cutoff value to discriminate clinically significant prostate cancer from benign prostate diseases in patients with suspected prostate cancer by 68Ga-PSMA PET/CT: a real-world study. Theranostics. 2021;11:8396-8411
94. Jafari E, Manafi-Farid R, Ahmadzadehfar H, Salek F, Jokar N, Keshavarz A. et al. Prognostic significance of baseline clinical and [68Ga]Ga-PSMA PET derived parameters on biochemical response, overall survival, and PSA progression-free survival in metastatic castration-resistant prostate cancer (mCRPC) patients undergoing [177Lu]Lu-PSMA therapy. Nuklearmedizin. 2024;63:347-358
95. Emmett L, Papa N, Subramaniam S, Crumbaker M, Nguyen A, Joshua AM. et al. Prognostic and predictive value of baseline PSMA-PET total tumour volume and SUVmean in metastatic castration-resistant prostate cancer in ENZA-p (ANZUP1901): a substudy from a multicentre, open-label, randomised, phase 2 trial. Lancet Oncol. 2025;26:1168-1177
96. Kimura K, Murthy V, Voter AF, Tuchayi AM, Yadav S, Solnes LB. et al. Assessment of PSMA PET/CT derived predictive markers for 177Lu-PSMA-617 treatment outcomes: results from the U.S. expanded-access program. J Clin Oncol. 2025;43(Suppl 16):5079
97. Gallium Ga 68 PSMA-11 Injection prescribing information. University of California, Los Angeles. 2020.
98. Gallium Ga 68 PSMA-11 Injection prescribing information. University of California, San Francisco. 2020.
99. Phillips R, Shi WY, Deek M, Radwan N, Lim SJ, Antonarakis ES. et al. Outcomes of observation vs stereotactic ablative radiation for oligometastatic prostate cancer: The ORIOLE phase 2 randomized clinical trial. JAMA Oncol. 2020;6:650-659
100. Guglielmo P, Buffi N, Porreca A, Setti L, Aricò D, Muraglia L. et al. Current insights on PSMA PET/CT in intermediate-risk prostate cancer: a literature review. Ann Nucl Med. 2025;39:247-254
101. Hamid Y, Rabbani RD, Afsara R, Nowrin S, Ghose A, Papadopoulos V. et al. Exosomal liquid biopsy in prostate cancer: a systematic review of biomarkers for diagnosis, prognosis, and treatment response. Int J Mol Sci. 2025;26(2):802
102. Van de Wiele C, Sathekge M, de Spiegeleer B, De Jonghe PJ, Debruyne PR, Borms M. et al. PSMA expression on neovasculature of solid tumors. Histol Histopathol. 2020;35:919-927
103. Sadaghiani MS, Baskaran S, Gorin MA, Rowe SP, Provost JC, Teslenko I. et al. Utility of PSMA PET/CT in staging and restaging of renal cell carcinoma: a systematic review and metaanalysis. J Nucl Med. 2024;65:1007-1012
104. Bertagna F, Albano D, Cerudelli E, Gazzilli M, Tomasini D, Bonù M. et al. Radiolabelled PSMA PET/CT in breast cancer. A systematic review. Nucl Med Rev Cent East Eur. 2020;23:32-35
105. van Lith SAM, Pruis IJ, Tolboom N, Snijders TJ, Henssen D, Ter Laan M. et al. PET imaging and protein expression of prostate-specific membrane antigen in glioblastoma: a multicenter inventory study. J Nucl Med. 2023;64:1526-1531
106. Dondi F, Albano D, Cerudelli E, Gazzilli M, Giubbini R, Treglia G. et al. Radiolabelled PSMA PET/CT or PET/MRI in hepatocellular carcinoma (HCC): a systematic review. Clin Transl Imaging. 2020;8:461-467
107. Jadvar H, Iravani A. Therapy strategies to defeat prostate cancer heterogeneity. Semin Nucl Med. 2026;56:18-21
Author contact
Corresponding author: Steven Eric Finkelstein, MD, DABR, FACRO, National Director of the Center of Advanced Radiation Excellence (CARE) and Director Radiation Oncology Research, Associated Medical Professionals / US Urology Partners, Chair of Board, The Luther Brady Foundation of the American College of Radiation Oncology (ACRO), Board of Chancellors, American College of Radiation Oncology (ACRO), 1226 East Water Street, Syracuse, New York 13210, Email: Steven.Finkelsteincom, Office 315-478-3468.
 Global reach, higher impact
Global reach, higher impact