13.3
Impact Factor
Theranostics 2023; 13(15):5151-5169. doi:10.7150/thno.85549 This issue Cite
Research Paper
PET imaging of focused-ultrasound enhanced delivery of AAVs into the murine brain
1. Molecular Imaging Program at Stanford (MIPS), Department of Radiology, School of Medicine, Stanford University, Stanford, CA, USA.
2. Stanford Cyclotron & Radiochemistry Facility (CRF), Department of Radiology, School of Medicine, Stanford University, Stanford, CA, USA.
* Authors contributed equally.
Abstract
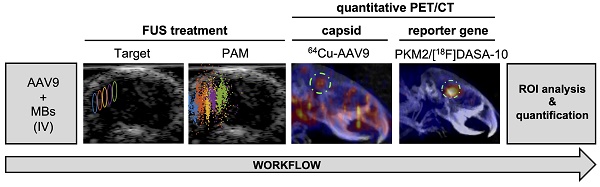
Rationale: Despite recent advances in the use of adeno-associated viruses (AAVs) as potential vehicles for genetic intervention of central and peripheral nervous system-associated disorders, gene therapy for the treatment of neuropathology in adults has not been approved to date. The currently FDA-approved AAV-vector based gene therapies rely on naturally occurring serotypes, such as AAV2 or AAV9, which display limited or no transport across the blood-brain barrier (BBB) if systemically administered. Recently developed engineered AAV variants have shown broad brain transduction and reduced off-target liver toxicity in non-human primates (NHPs). However, these vectors lack spatial selectivity for targeted gene delivery, a potentially critical limitation for delivering therapeutic doses in defined areas of the brain. The use of microbubbles, in conjunction with focused ultrasound (FUS), can enhance regional brain AAV transduction, but methods to assess transduction in vivo are needed.
Methods: In a murine model, we combined positron emission tomography (PET) and optical imaging of reporter gene payloads to non-invasively assess the spatial distribution and transduction efficiency of systemically administered AAV9 after FUS and microbubble treatment. Capsid and reporter probe accumulation are reported as percent injected dose per cubic centimeter (%ID/cc) for in vivo PET quantification, whereas results for ex vivo assays are reported as percent injected dose per gram (%ID/g).
Results: In a study spanning accumulation and transduction, mean AAV9 accumulation within the brain was 0.29 %ID/cc without FUS, whereas in the insonified region of interest of FUS-treated mice, the spatial mean and maximum reached ~2.3 %ID/cc and 4.3 %ID/cc, respectively. Transgene expression assessed in vivo by PET reporter gene imaging employing the pyruvate kinase M2 (PKM2)/[18F]DASA-10 reporter system increased up to 10-fold in the FUS-treated regions, as compared to mice receiving AAVs without FUS. Systemic injection of AAV9 packaging the EF1A-PKM2 transgene followed by FUS in one hemisphere resulted in 1) an average 102-fold increase in PKM2 mRNA concentration compared to mice treated with AAVs only and 2) a 12.5-fold increase in the insonified compared to the contralateral hemisphere of FUS-treated mice.
Conclusion: Combining microbubbles with US-guided treatment facilitated a multi-hour BBB disruption and stable AAV transduction in targeted areas of the murine brain. This unique platform has the potential to provide insight and aid in the translation of AAV-based therapies for the treatment of neuropathologies.
Keywords: Gene therapy, Adeno-associated virus, Focused ultrasound, Blood-brain barrier, Positron emission tomography
 Global reach, higher impact
Global reach, higher impact