13.3
Impact Factor
Theranostics 2023; 13(8):2588-2604. doi:10.7150/thno.83817 This issue Cite
Research Paper
Inactivating IL34 promotes regenerating muscle stem cell expansion and attenuates Duchenne muscular dystrophy in mouse models
1. State Key Laboratories for Agrobiotechnology, College of Biological Sciences, China Agricultural University, Yuanmingyuan West Road No. 2, Haidian District, Beijing 100193, China.
2. Department of Cell Biology, Third Military Medical University (Army Medical University), Gaotanyan Road No. 30, Shapingba District, Chongqing 400038, China.
3. State Key Lab of Animal Nutrition, College of Animal Science and Technology, China Agricultural University, Yuanmingyuan West Road No. 2, Haidian District, Beijing 100193, China.
4. Beijing Advanced Innovation Center for Food Nutrition and Human Health, College of Biological Sciences, China Agricultural University, Yuanmingyuan West Road No. 2, Haidian District, Beijing 100193, China.
# Yang Su and Yuxin Cao contributed equally to this work.
Abstract
Background: The balance between the differentiation and self-renewal of satellite cells (SCs) is essential for skeletal muscle homeostasis and regeneration. Our knowledge of this regulatory process is incomplete.
Methods: Using global and conditional knockout mice as in vivo models and isolated satellite cells as in vitro system, we investigated the regulatory mechanisms of IL34 in the process of skeletal muscle regeneration in vivo and in vitro.
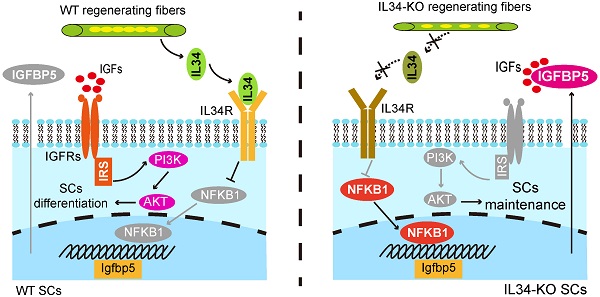
Results: Myocytes and regenerating fibers are major source of IL34. Deletion of interleukin 34 (IL34) sustains expansion by sacrificing the differentiation of SCs and leads to significant muscle regeneration defects. We further found that inactivating IL34 in SCs leads to hyperactivation of NFKB1 signaling; NFKB1 translocates to the nucleus and binds to the promoter region of Igfbp5 to synergistically disturb protein kinase B (Akt) activity. Notably, augmented Igfbp5 function in SCs led to deficient differentiation and Akt activity. Furthermore, disrupting Akt activity both in vivo and in vitro mimicked the phenotype of IL34 knockout. Finally, deleting IL34 or interfering Akt in mdx mice ameliorates dystrophic muscles.
Conclusion: We comprehensively characterized regenerating myofibers-expressed IL34 plays a pivotal role in controlling myonuclear domain. The results also indicate that impairing IL34 function by promoting SC maintenance can lead to improved muscular performance in mdx mice in which the stem cell pool is compromised.
Keywords: IL34, satellite cells, muscle regeneration, NFKB1, DMD
 Global reach, higher impact
Global reach, higher impact