13.3
Impact Factor
Theranostics 2023; 13(7):2263-2280. doi:10.7150/thno.83340 This issue Cite
Research Paper
OTUD1 promotes pathological cardiac remodeling and heart failure by targeting STAT3 in cardiomyocytes
1. Department of Pharmacy and Institute of Inflammation, Zhejiang Provincial People's Hospital, Affiliated People's Hospital, Hangzhou Medical College, Hangzhou, Zhejiang, 310014, China.
2. Chemical Biology Research Center, School of Pharmaceutical Sciences, Wenzhou Medical University, Wenzhou, Zhejiang, 325035, China.
3. Key Laboratory of Natural Medicines of the Changbai Mountain, Ministry of Education, Yanbian University, Yanji 133002, China.
4. Department of Cardiology, the First Affiliated Hospital of Wenzhou Medical University, Wenzhou, Zhejiang, 325035, China.
5. Department of Ultrasonography, the Second Affiliated Hospital, Wenzhou Medical University, Wenzhou, Zhejiang, 325000, China.
Abstract
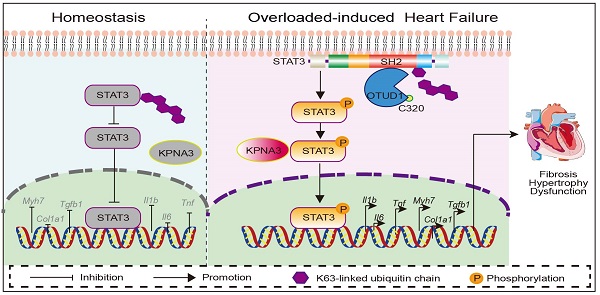
Rationale: Understanding the molecular mechanisms of deleterious cardiac remodeling is important for the development of treatments for heart failure. Recent studies have highlighted a role of deubiquitinating enzymes in cardiac pathophysiology. In the present study, we screened for alteration of deubiquitinating enzymes in experimental models of cardiac remodeling, which indicated a potential role of OTU Domain-Containing Protein 1 (OTUD1).
Methods: Wide-type or OTUD1 knockout mice with chronic angiotensin II infusion and transverse aortic constriction (TAC) were utilized to develop cardiac remodeling and heart failure. We also overexpressed OTUD1 in mouse heart with AAV9 vector to validate the function of OTUD1. LC-MS/MS analysis combined with Co-IP was used to identify the interacting proteins and substrates of OTUD1.
Results: We found that OTUD1 is elevated in mouse heart tissues following chronic angiotensin II administration. OTUD1 knockout mice were significantly protected against angiotensin II-induced cardiac dysfunction, hypertrophy, fibrosis and inflammatory response. Similar results were obtained in the TAC model. Mechanistically, OTUD1 bounds to the SH2 domain of STAT3 and causes deubiquitination of STAT3. Cysteine at position 320 of OTUD1 exerts K63 deubiquitination to promote STAT3 phosphorylation and nuclear translocation, thereby increasing STAT3 activity to induce inflammatory responses, fibrosis, and hypertrophy in cardiomyocytes. Finally, OTUD1 overexpression by AAV9 vector increases Ang II-induced cardiac remodeling in mice and OTUD1-regulated responses can be inhibited by blocking STAT3.
Conclusion: Cardiomyocyte OTUD1 promotes pathological cardiac remodeling and dysfunction by deubiquitinating STAT3. These studies have highlighted a novel role of OTUD1 in hypertensive heart failure and identified STAT3 as a target of OTUD1 in mediating these actions.
Keywords: OTUD1, Angiotensin II, Deubiquitination enzyme, STAT3, Heart failure
 Global reach, higher impact
Global reach, higher impact