Impact Factor
Theranostics 2023; 13(4):1247-1263. doi:10.7150/thno.80294 This issue Cite
Research Paper
Proteomes from AMPK-inhibited peripheral blood mononuclear cells suppress the progression of breast cancer and bone metastasis
1. Department of Pharmacology, School of Pharmacy, Harbin Medical University, Harbin 150081, China
2. Department of Biomedical Engineering, Indiana University Purdue University Indianapolis, Indianapolis, IN 46202, USA
3. Department of Radiation Oncology, Osaka University Graduate School of Medicine; Suita, Osaka 565-0871, Japan
4. Indiana Center for Musculoskeletal Health, Indiana University School of Medicine, Indianapolis, IN 46202, USA
5. Simon Comprehensive Cancer Center, Indiana University School of Medicine, Indianapolis, IN 46202, USA
Abstract
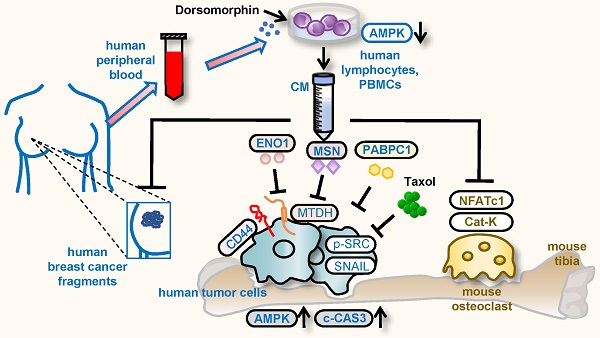
Background: During a developmental process, embryos employ varying tactics to remove unwanted cells. Using a procedure analogous to some of the embryonic cells, we generated a tumor-eliminating conditioned medium (CM) from AMPK-inhibited lymphocytes and monocytes in peripheral blood mononuclear cells (PBMCs).
Methods: AMPK signaling was inhibited by the application of a pharmacological agent, Dorsomorphin, and the therapeutic effects of their conditioned medium (CM) were evaluated using in vitro cell cultures, ex vivo breast cancer tissues, and a mouse model of mammary tumors and tumor-induced osteolysis. The regulatory mechanism was evaluated using mass spectrometry-based proteomics, Western blotting, immunoprecipitation, gene overexpression, and RNA interference.
Results: While AMPK signaling acted mostly anti-tumorigenic, we paradoxically inhibited it to build induced tumor-suppressing cells and their tumor-eliminating CM. In a mouse model of breast cancer, the application of AMPK-inhibited lymphocyte-derived CM reduced mammary tumors additively to a chemotherapeutic agent, Taxol. It also prevented bone loss in the tumor-bearing tibia. Furthermore, the application of CM from the patient-derived peripheral blood diminished ex vivo breast cancer tissues isolated from the same patients. Notably, proteins enriched in CM included Moesin (MSN), Enolase 1 (ENO1), and polyA-binding protein 1 (PABPC1), which are considered tumorigenic in many types of cancer. The tumor-suppressing actions of MSN and ENO1 were at least in part mediated by Metadherin (Mtdh), which is known to promote metastatic seeding.
Conclusion: We demonstrated that PBMCs can be used to generate tumor-suppressive proteomes, and extracellular tumor-suppressing proteins such as MSN, ENO1, and PABPC1 are converted from tumor-promoting factors inside cancer cells. The results support the possibility of developing autologous blood-based therapy, in which tumor-suppressing proteins are enriched in engineered PBMC-derived CM by the inhibition of AMPK signaling.
Keywords: breast cancer bone metastasis, PBMCs, conditioned medium, AMPK, Metadherin
 Global reach, higher impact
Global reach, higher impact