Impact Factor
Theranostics 2023; 13(2):472-482. doi:10.7150/thno.79452 This issue Cite
Research Paper
Simplified one-pot 18F-labeling of biomolecules with in situ generated fluorothiophosphate synthons in high molar activity
1. Center for Molecular Imaging and Translational Medicine, State Key Laboratory of Molecular Vaccinology and Molecular Diagnostics, Department of Laboratory Medicine, School of Public Heath, Xiamen University, Xiamen, Fujian 361102, China.
2. Tianjin Engineering Technology Center of Chemical Wastewater Source Reduction and Recycling, School of Science, Tianjin Chengjian University, Tianjin 300384, China.
3. School of Pharmaceutical Sciences, Xiamen University, Xiamen, Fujian 361102, China.
Abstract
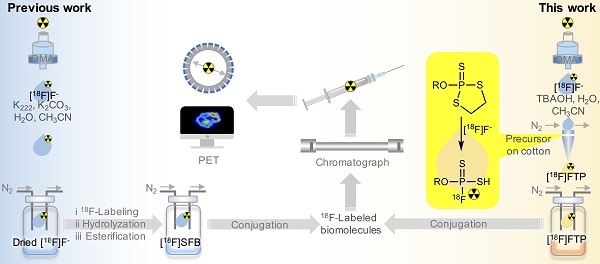
Rationale: Conventional 18F-labeling methods that demand substrate pre-modification or lengthy radiosynthesis procedures have impeded the visualization and translation of numerous biomolecules, as biomarkers or ligands, using modern positron emission tomography techniques in vivo. Moreover, 18F-labeled biomolecules in high molar activity (Am) that are indispensable for sensitive imaging could be only achieved under strict labeling conditions.
Methods: Herein, 18F-labeled fluorothiophosphate (FTP) synthons in high Am have been generated rapidly in situ in reaction solutions with < 5% water via nucleophilic substitution by wet [18F]F-, which required minimal processing from cyclotron target water.
Results: Various 18F-labeled FTP synthons have been prepared in 30 sec at room temperature with high radiochemical yields > 75% (isolated, non-decay-corrected). FTP synthons with unsaturated hydrocarbon or activated ester group can conjugate with typical small molecules, peptides, proteins, and metallic nanoparticles. 337-517 GBq μmol-1 Am has been achieved for 18F-labeled c(RGDyK) peptide using an automatic module with 37-74 GBq initial activity.
Conclusion: The combination of high 18F-fluorination efficiency of FTP synthons and following mild conjugation condition provides a universal simplified one-pot 18F-labeling method for broad unmodified biomolecular substrates.
Keywords: radiolabeling, radiosynthon, fluorine-18, fluorothiophosphate, positron emission tomography probe
 Global reach, higher impact
Global reach, higher impact