Impact Factor
Theranostics 2022; 12(8):3995-4009. doi:10.7150/thno.65235 This issue Cite
Research Paper
Lactate metabolism coordinates macrophage response and regeneration in zebrafish
1. IRMB, Univ Montpellier, INSERM, Montpellier, France
2. CHU Montpellier, Montpellier, F-34295 France
* Equally contributing authors
# Equally contributing authors
Abstract
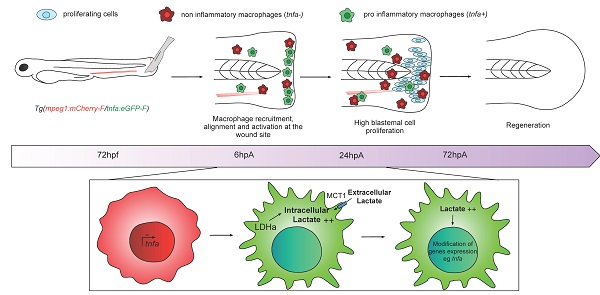
Rationale: Macrophages are multifunctional cells with a pivotal role on tissue development, homeostasis and regeneration. Indeed, in response to tissue injury and the ensuing regeneration process, macrophages are challenged and undergo massive metabolic adaptations and changes. However, the control of this metabolic reprogramming by macrophage microenvironment has never been deciphered in vivo.
Methods: In this study, we used zebrafish model and caudal fin resection as a robust regeneration system. We explored specific changes in gene expression after tissue amputation via single-cell RNA sequencing analysis and whole-tissue transcriptomic analysis. Based on the identification of key modifications, we confirmed the role of the lactate pathway in macrophage response and fin regeneration, through the combination of chemical and genetic inhibitors of this pathway.
Results: Single cell RNA sequencing revealed the upregulation of different genes associated with glycolysis and lactate metabolism in macrophages, upon fin regeneration. Hence, using chemical inhibitors of the LDH enzyme, we confirmed the role of lactate in macrophage recruitment and polarization, to promote a pro-inflammatory phenotype and enhance fin regeneration. The genetic modulation of monocarboxylate transporters illustrated a complex regulation of lactate levels, based on both intracellular and extracellular supplies. Commonly, the different sources of lactate resulted in macrophage activation with an increased expression level of inflammatory cytokines such as TNFa during the first 24 hours of regeneration. Transcriptomic analyses confirmed that lactate induced a global modification of gene expression in macrophages.
Conclusion: Altogether, our findings highlight the crucial role of lactate at the onset of macrophage differentiation toward a pro-inflammatory phenotype. The deep modifications of macrophage phenotype mediated by lactate and downstream effectors play a key role to coordinate inflammatory response and tissue regeneration.
Keywords: Regeneration, lactate, macrophage, zebrafish, single cell RNA-sequencing
 Global reach, higher impact
Global reach, higher impact