Impact Factor
Theranostics 2022; 12(8):3963-3976. doi:10.7150/thno.70893 This issue Cite
Research Paper
Development of human cartilage circadian rhythm in a stem cell-chondrogenesis model
1. Wellcome Centre for Cell Matrix Research, Faculty of Biology, Medicine and Health, University of Manchester, Oxford Road, Manchester, UK
2. Division of Cell Matrix Biology and Regenerative Medicine, School of Biological Sciences, Faculty of Biology, Medicine and Health, University of Manchester, Manchester Academic Health Science Centre, Manchester, UK
3. Bioinformatics Core Facility, Faculty of Biology, Medicine and Health, University of Manchester, Oxford Road, Manchester, UK
4. Department of Biomedical Engineering, The Hong Kong Polytechnic University, Hung Hom, Hong Kong, China
Abstract
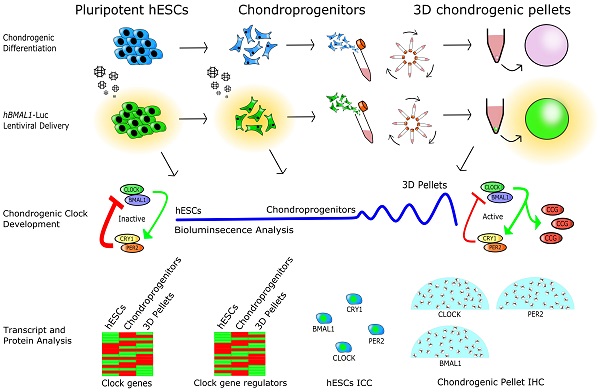
The circadian clock in murine articular cartilage is a critical temporal regulatory mechanism for tissue homeostasis and osteoarthritis. However, translation of these findings into humans has been hampered by the difficulty in obtaining circadian time series human cartilage tissues. As such, a suitable model is needed to understand the initiation and regulation of circadian rhythms in human cartilage.
Methods: We used a chondrogenic differentiation protocol on human embryonic stem cells (hESCs) as a proxy for early human chondrocyte development. Chondrogenesis was validated using histology and expression of pluripotency and differentiation markers. The molecular circadian clock was tracked in real time by lentiviral transduction of human clock gene luciferase reporters. Differentiation-coupled gene expression was assessed by RNAseq and differential expression analysis.
Results: hESCs lacked functional circadian rhythms in clock gene expression. During chondrogenic differentiation, there was an expected reduction of pluripotency markers (e.g., NANOG and OCT4) and a significant increase of chondrogenic genes (SOX9, COL2A1 and ACAN). Histology of the 3D cartilage pellets at day 21 showed a matrix architecture resembling human cartilage, with readily detectable core clock proteins (BMAL1, CLOCK and PER2). Importantly, the circadian clocks in differentiating hESCs were activated between day 11 (end of the 2D stage) and day 21 (10 days after 3D differentiation) in the chondrogenic differentiation protocol. RNA sequencing revealed striking differentiation coupled changes in the expression levels of most clock genes and a range of clock regulators.
Conclusions: The circadian clock is gradually activated through a differentiation-coupled mechanism in a human chondrogenesis model. These findings provide a human 3D chondrogenic model to investigate the role of the circadian clock during normal homeostasis and in diseases such as osteoarthritis.
Keywords: Human stem cells, chondrogenesis, circadian rhythm, development, osteoarthritis
 Global reach, higher impact
Global reach, higher impact