Impact Factor
Theranostics 2022; 12(8):3882-3895. doi:10.7150/thno.70830 This issue Cite
Research Paper
Selenium nanoparticles alleviate ischemia reperfusion injury-induced acute kidney injury by modulating GPx-1/NLRP3/Caspase-1 pathway
1. Department of Nephrology, The Key Laboratory for The Prevention and Treatment of Chronic Kidney Disease of Chongqing, Xinqiao Hospital, Army Medical University (Third Military Medical University), Chongqing, 400037, China.
2. State Key Laboratory of Trauma, Burns and Combined Injury, Institute of Combined Injury of PLA, Chongqing Engineering Research Center for Nanomedicine, College of Preventive Medicine, Army Medical University (Third Military Medical University), Chongqing, 400038, China.
3. Biomedical Analysis Center, Army Medical University (Third Military Medical University), Chongqing, 400038, China.
#These authors contributed equally to this work.
Abstract
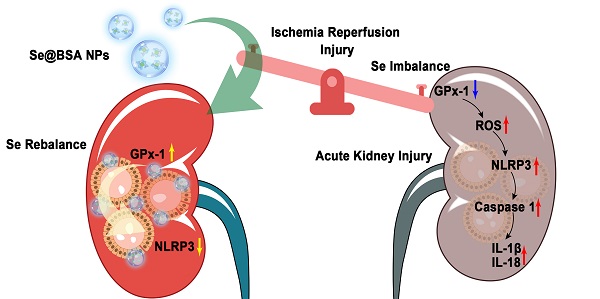
Rationale: Acute kidney injury (AKI) is a common critical illness in the clinic and currently lacks effective treatment options. Ischemia reperfusion injury (IRI) is a major pathogenic factor for AKI. Due to the deficiency of selenium (Se) in AKI patients, we intended to treat IRI-induced AKI using a Se rebalancing strategy in the present study.
Methods: Sodium selenate, ascorbic acid, and bovine serum albumin (BSA) were employed to prepare nanomaterials termed Se@BSA nanoparticles (NPs) using a simple method. Experiments with human renal tubular epithelial HK-2 cells exposed to hypoxia/reoxygenation (H/R) and IRI-AKI mice were used to evaluate the therapeutic efficiency of Se@BSA NPs. Transcriptome sequencing, further molecular biology experiments, and pathologic analysis were performed to investigate the underlying mechanisms.
Results: Se@BSA NPs accumulated in mouse kidneys and could be endocytosed by renal tubular epithelial cells after intravenous administration. In vitro studies showed that Se@BSA NP treatment markedly increased the levels of glutathione peroxidase (GPx)-1 and suppressed NLRP3 inflammasome activation in H/R cells, which resulted in reductions in the proteolytic cleavage of pro-Caspase-1 into active Caspase-1 and the maturation of inflammatory factors. Mouse experiments confirmed these findings and demonstrated an inspiring mitigative effect of Se@BSA NPs on IRI-induced AKI. Owing to modulation of the GPx-1/NLRP3/Caspase-1 pathway, Se@BSA NPs dramatically inhibited fibrosis formation after AKI.
Conclusion: This study provides an effective therapeutic option by applying easy-to-produce Se-containing nanomaterials to remedy Se imbalance and impede inflammatory responses in the kidney, which is a promising candidate for AKI treatment.
Keywords: acute kidney injury, ischemia reperfusion injury, selenium nanoparticles, glutathione peroxidase-1, inflammasome
 Global reach, higher impact
Global reach, higher impact