Impact Factor
Theranostics 2022; 12(8):3834-3846. doi:10.7150/thno.70668 This issue Cite
Research Paper
Orally administered titanium carbide nanosheets as anti-inflammatory therapy for colitis
1. Institute of Functional Nano & Soft Materials (FUNSOM), Jiangsu Key Laboratory for Carbon-Based Functional Materials & Devices, Soochow University, Suzhou 215123, China
2. College of Science, State Key Laboratory of Agricultural Microbiology, Huazhong Agricultural University, Wuhan, 430070, China
3. Department of Gastrointestinal Surgery, The Second Affiliated Hospital, Wenzhou Medical University, Wenzhou, Zhejiang Province, China
Abstract
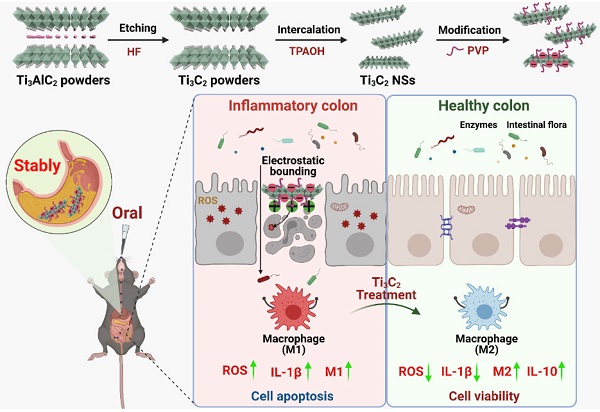
Rationale: Oxidative stress, resulting from excessive reactive oxygen species (ROS), plays an important role in the initiation and progression of inflammatory bowel disease (IBD). Therefore, developing novel strategies to target the disease location and treat inflammation is urgently needed.
Methods: Herein, we designed and developed a novel and effective antioxidant orally-administered nanoplatform based on simulated gastric fluid (SGF)-stabilized titanium carbide MXene nanosheets (Ti3C2 NSs) with excellent biosafety and multiple ROS-scavenging abilities for IBD therapy.
Results: This broad-spectrum and efficient ROS scavenging performance was mainly relied on the strong reducibility of Ti-C bound. Intracellular ROS levels confirmed that Ti3C2 NSs could efficiently eliminate excess ROS against oxidative stress-induced cell damage. Following oral administration, negatively-charged Ti3C2 NSs specifically adsorbed onto the positively-charged inflamed colon tissue via electrostatic interaction, leading to efficient therapy of dextran sulfate sodium salt (DSS)-induced colitis. The therapeutic mechanism mainly attributed to decreased ROS levels and pro-inflammatory cytokine secretion, and increased M2-phenotype macrophage infiltration and anti-inflammatory cytokine secretion, efficiently inhibiting inflammation and alleviating colitis symptoms. Due to their excellent ROS-scavenging performance, Ti3C2-based woundplast also promoted skin wound healing and functional vessel formation.
Conclusions: Our study introduces redox-mediated antioxidant MXene nanoplatform as a novel type of orally administered nanoagents for treating IBD and other inflammatory diseases of the digestive tract.
Keywords: Ti3C2 nanosheets, ROS scavenging, oral administration, anti-inflammation, IBD therapy
 Global reach, higher impact
Global reach, higher impact