Impact Factor
Theranostics 2022; 12(8):3656-3675. doi:10.7150/thno.70370 This issue Cite
Research Paper
Requirement of hippocampal DG nNOS-CAPON dissociation for the anxiolytic and antidepressant effects of fluoxetine
1. Key Laboratory of Developmental Genes and Human Diseases, MOE, Department of Histology and Embryology, School of Medicine, Southeast University, Nanjing, Jiangsu 210009, PR China.
2. Department of Pharmacy, The Affiliated Jiangyin Hospital, School of Medicine, Southeast University, Jiangyin, Jiangsu 214400, PR China.
3. Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, Shanghai, 201108, China.
#These authors contributed equally to this work.
Abstract
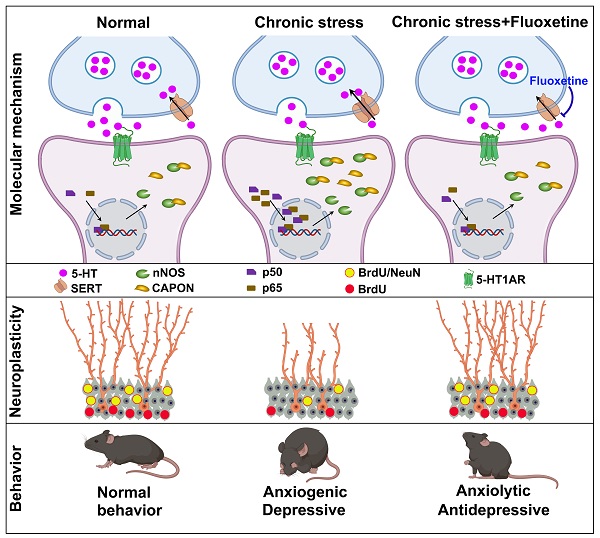
Background: Adult hippocampal neurogenesis and synaptic plasticity are necessary for the behavioral response to the selective serotonin reuptake inhibitor (SSRI) fluoxetine, but the molecular mechanisms underlying these effects are only partially understood.
Methods: Anxiety and depressive-like behaviors in mice were developed by chronic mild stress (CMS) or chronic corticosterone (CORT) treatment. Pharmacological and genetic approaches were used to investigate the role of the neuronal nitric oxide synthase (nNOS)-carboxy-terminal PDZ ligand of nNOS (CAPON) interaction in behavioral and neuroplasticity effects of serotoninergic system. Molecular biological and morphological studies were performed to examine the mechanisms underlying the behavioral effects of nNOS-CAPON interaction that modulated by 5-HT1A receptor (5-HT1AR).
Results: Fluoxetine prevented chronic stress-induced nNOS-CAPON upregulation and coupling in the dentate gyrus (DG), and promoting nNOS-CAPON association weakened the anxiolytic and antidepressant effects of fluoxetine in stressed mice. The chronic fluoxetine elevated 5-HT and 5HT1AR agonist 8-OH-DPAT decreased the expression and binding of nNOS with CAPON, whereas 5-HT1AR antagonist NAN-190 had the opposite effects. Importantly, augmenting nNOS-CAPON binding neutralized 8-OH-DPAT-upregulated spine density of DG granule cells and well-characterized synaptic-related proteins, including brain-derived neurotrophic factor (BDNF) and phosphorylation of extracellular signal regulated kinase (ERK), cAMP-response element binding protein (CREB), and synapsin in the DG and abolished the anxiolytic and antidepressant-like effects of 8-OH-DPAT. In contrast, dissociation of nNOS from CAPON rescued the effects of NAN-190 on behavior and neuroplasticity.
Conclusion: Taken together, our results indicated that fluoxetine modifies mood behaviors and hippocampal neuroplasticity by disrupting the nNOS-CAPON interaction that links postsynaptic 5-HT1AR activation.
Keywords: anxiety, depression, fluoxetine, 5-HT1A receptor, nNOS-CAPON coupling
 Global reach, higher impact
Global reach, higher impact