13.3
Impact Factor
Theranostics 2022; 12(7):3456-3473. doi:10.7150/thno.71129 This issue Cite
Research Paper
Polymer chimera of stapled oncolytic peptide coupled with anti-PD-L1 peptide boosts immunotherapy of colorectal cancer
1. Shanghai Frontiers Science Center for Chinese Medicine Chemical Biology, Institute of Interdisciplinary Integrative Medicine Research, Shanghai University of Traditional Chinese Medicine, Shanghai, 201203, China
2. School of Pharmacy, Fudan University, Shanghai, 201203, China.
3. Department of Chemistry and Biochemistry, College of Liberal Arts, University of Mississippi, University, MS, 38677-1848, USA
4. Department of BioMolecular Sciences and Research Institute of Pharmaceutical Sciences, School of Pharmacy, University of Mississippi, University, MS 38677-1848, USA
*These authors contributed equally to this work.
Abstract
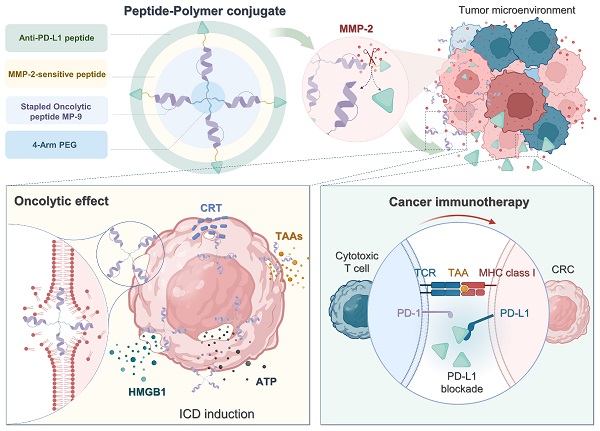
Rationale: Scarce tumor mutation burden and neoantigens create tremendous obstacles for an effective immunotherapy of colorectal cancer (CRC). Oncolytic peptides rise as a promising therapeutic approach that boosts tumor-specific immune responses by inducing antigenic substances. However, the clinical application of oncolytic peptides has been hindered because of structural instability, proteolytic degradation, and undesired toxicity when administered systemically.
Methods: Based on wasp venom peptide, an optimized stapled oncolytic peptide MP9 was developed with rigid α-helix, protease-resistance, and CRC cell cytotoxicity. By incorporating four functional motifs that include D-peptidomimetic inhibitor of PD-L1, matrix metalloproteinase-2 (MMP-2) cleavable spacer, and MP9 with 4-arm PEG, a novel peptide-polymer conjugate (PEG-MP9-aPDL1) was obtained and identified as the most promising systemic delivery vehicle with PD-L1 targeting specificity and favorable pharmacokinetic properties.
Results: We demonstrated that PEG-MP9-aPDL1-driven oncolysis induces a panel of immunogenic cell death (ICD)-relevant damage-associated molecular patterns (DAMPs) both in vitro and in vivo, which are key elements for immunotherapy with PD-L1 inhibitor. Further, PEG-MP9-aPDL1 exhibited prominent immunotherapeutic efficacy in a CRC mouse model characterized by tumor infiltration of CD8+ T cells and induction of cytotoxic lymphocytes (CTLs) in the spleens.
Conclusion: Our findings suggest that PEG-MP9-aPDL1 is an all-in-one platform for oncolytic immunotherapy and immune checkpoint blockade (ICB).
Keywords: colorectal cancer, stapled mastoparan peptide, peptide-polymer conjugate, immunogenic cell death, oncolytic immunotherapy
 Global reach, higher impact
Global reach, higher impact