13.3
Impact Factor
Theranostics 2022; 12(7):3420-3437. doi:10.7150/thno.70346 This issue Cite
Research Paper
Intelligent design of polymer nanogels for full-process sensitized radiotherapy and dual-mode computed tomography/magnetic resonance imaging of tumors
1. State Key Laboratory for Modification of Chemical Fibers and Polymer Materials, Shanghai Engineering Research Center of Nano-Biomaterials and Regenerative Medicine, College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, P. R. China
2. Department of Radiation Oncology, Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200080, P. R. China
3. DWI-Leibniz-Institute for Interactive Materials e.V., Forckenbeckstr. 50, 52056, Aachen, Germany
4. Functional and Interactive Polymers, Institute of Technical and Macromolecular Chemistry, RWTH Aachen University, Worringerweg 2, 52074, Aachen, Germany
5. Department of Nuclear Medicine, Shanghai Cancer Center, Fudan University, Shanghai 200030, P. R. China
*Authors contributed equally to this work.
Abstract
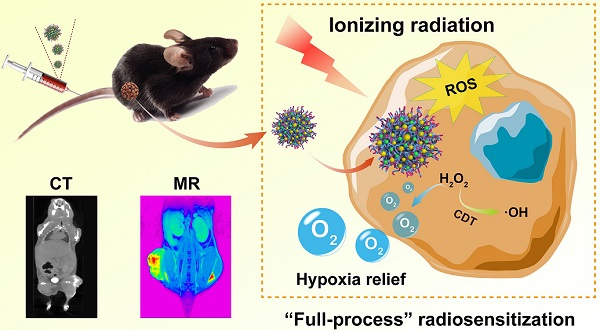
Rationale: Development of intelligent radiosensitization nanoplatforms for imaging-guided tumor radiotherapy (RT) remains challenging. We report here the construction of an intelligent nanoplatform based on poly(N-vinylcaprolactam) (PVCL) nanogels (NGs) co-loaded with gold (Au) and manganese dioxide (MnO2) nanoparticles (NPs) for dual-mode computed tomography (CT)/magnetic resonance (MR) imaging-guided “full-process” sensitized RT of tumors.
Methods: PVCL NGs were synthesized via precipitation polymerization and in situ loaded with Au and MnO2 NPs. The created PVCL-Au-MnO2 NGs were well characterized and systematically examined in their cytotoxicity, cellular uptake, intracellular oxygen and ·OH production, and cell cycle arrest in vitro, evaluated to disclose their RT sensitization effects of cancer cells and a tumor model, and assessed to validate their dual-mode CT/MR imaging potential, pharmacokinetics, biodistribution, and biosafety in vivo.
Results: The formed PVCL-Au-MnO2 NGs with a size of 121.5 nm and good stability can efficiently generate reactive oxygen species through a Fenton-like reaction to result in cell cycle distribution toward highly radiosensitive G2/M phase prior to X-ray irradiation, sensitize the RT of cancer cells under X-ray through the loaded Au NPs to induce the significant DNA damage, and further prevent DNA-repairing process after RT through the continuous production of O2 catalyzed by MnO2 in the hybrid NGs to relieve the tumor hypoxia. Likewise, the in vivo tumor RT can also be guided through dual mode CT/MR imaging due to the Au NPs and Mn(II) transformed from MnO2 NPs.
Conclusion: Our study suggests an intelligent PVCL-based theranostic NG platform that can achieve “full-process” sensitized tumor RT under the guidance of dual-mode CT/MR imaging.
Keywords: Hybrid PVCL nanogels, Fenton-like reaction, manganese dioxide, tumor radiotherapy, full-process sensitization
 Global reach, higher impact
Global reach, higher impact