13.3
Impact Factor
Theranostics 2022; 12(7):3407-3419. doi:10.7150/thno.70701 This issue Cite
Research Paper
Phosphorus dendron nanomicelles as a platform for combination anti-inflammatory and antioxidative therapy of acute lung injury
1. State Key Laboratory for Modification of Chemical Fibers and Polymer Materials, Shanghai Engineering Research Center of Nano-Biomaterials and Regenerative Medicine, College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, People's Republic of China
2. Laboratoire de Chimie de Coordination du CNRS, 205 Route de Narbonne, BP 44099, 31077 Toulouse CEDEX 4, France
3. Université de Toulouse, UPS, INPT, 31077 Toulouse CEDEX 4, France
4. Université Paris Descartes, PRES Sorbonne Paris Cité, CNRS UMR 860, Laboratoire de Chimie et de Biochimie Pharmacologiques et Toxicologique, 45, rue des Saints Pères, 75006 Paris, France
5. CQM-Centro de Quimica da Madeira, Universidade da Madeira, 9020-105 Funchal, Portugal
Abstract
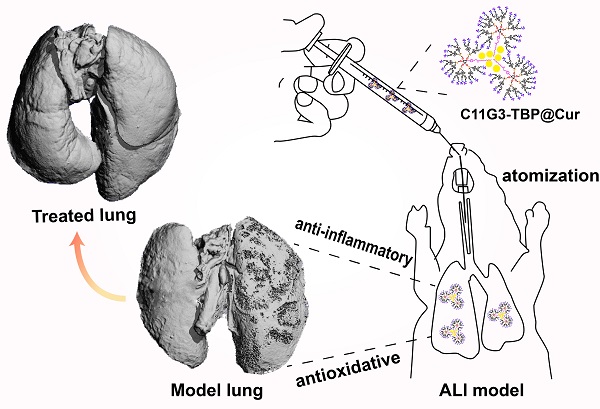
Rationale: Development of novel nanomedicines to inhibit pro-inflammatory cytokine expression and reactive oxygen species (ROS) generation for anti-inflammatory therapy of acute lung injury (ALI) remains challenging. Here, we present a new nanomedicine platform based on tyramine-bearing two dimethylphosphonate sodium salt (TBP)-modified amphiphilic phosphorus dendron (C11G3) nanomicelles encapsulated with antioxidant drug curcumin (Cur).
Methods: C11G3-TBP dendrons were synthesized via divergent synthesis and self-assembled to generate nanomicelles in a water environment to load hydrophobic drug Cur. The created C11G3-TBP@Cur nanomicelles were well characterized and systematically examined in their cytotoxicity, cellular uptake, intracellular ROS elimination, pro-inflammatory cytokine inhibition and alveolar macrophages M2 type repolarization in vitro, and evaluated to assay their anti-inflammatory and antioxidative therapy effects of ALI mice model through pro-inflammatory cytokine expression level in bronchoalveolar lavage fluid and lung tissue, histological analysis and micro-CT imaging detection of lung tissue injury in vivo.
Results: The nanomicelles with rigid phosphorous dendron structure enable high-capacity and stable Cur loading. Very strikingly, the drug-free C11G3-TBP micelles exhibit excellent cytocompatibility and intrinsic anti-inflammatory activity through inhibition of nuclear transcription factor-kappa B, thus causing repolarization of alveolar macrophages from M1 type to anti-inflammatory M2 type. Taken together with the strong ROS scavenging property of the encapsulated Cur, the developed nanomicelles enable effective therapy of inflammatory alveolar macrophages in vitro and an ALI mouse model in vivo after atomization administration.
Conclusion: The created phosphorus dendron nanomicelles can be developed as a general nanomedicine platform for combination anti-inflammatory and antioxidative therapy of inflammatory diseases.
Keywords: phosphorus dendrons, nanomicelles, curcumin, alveolar macrophages, NF-κB, acute lung injury
 Global reach, higher impact
Global reach, higher impact