13.3
Impact Factor
Theranostics 2022; 12(7):3316-3328. doi:10.7150/thno.69684 This issue Cite
Research Paper
NAD+-boosting molecules suppress mast cell degranulation and anaphylactic responses in mice
1. Korea Zoonosis Research Institute, Chonbuk National University, Iksan 54596, Republic of Korea.
2. Department of Biochemistry and Molecular Biology, Chonbuk National University Medical School, Jeonju 54896, Republic of Korea.
3. Department of Obstetrics and Gynecology, Chonbuk National University Medical School, Jeonju 54896, Republic of Korea.
4. College of Pharmacy, Chonbuk National University, Jeonju 54896, Republic of Korea.
* These authors equally contributed to this work.
Abstract
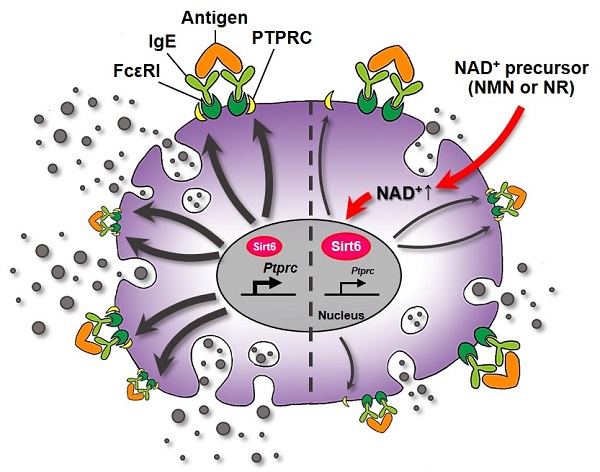
Nicotinamide adenine dinucleotide (NAD+) acts as a cofactor for multiple biological processes. While previous research has revealed that the NAD+ declines associated with aging contributes to an impairment of immune cells, its role in mast cell function, especially in response to an anaphylactic condition, has remained unexplored. We tested whether the restoration of cellular NAD+ concentration by the supplementation of NAD+ boosting molecules prevented mast cell degranulation and anaphylactic responses.
Methods: Bone marrow derived mast cells (BMMCs) and human cord blood derived mast cells were treated with NAD+ precursors nicotinamide mononucleotide (NMN) and nicotinamide riboside (NR), and FcεRI downstream signaling was assessed. Animal models of passive systemic anaphylaxis (PSA) and passive cutaneous anaphylaxis (PCA) were used to investigate the effects of NAD+ precursors in the anaphylactic responses of mice.
Results: Treatment of murine BMMCs and human cord blood derived mast cells with NAD+ precursors repressed intracellular signaling downstream of FcεRI, as well as the release of inflammatory cytokines and lipid mediators. The intraperitoneal administration of NMN or NR also markedly attenuated IgE-mediated anaphylactic responses in mouse models of PSA and PCA. These beneficial effects of NAD+ precursors, however, were attenuated in mast cell-specific Sirt6 knockout mice, indicating a Sirt6 dependency for their action.
Conclusion: NAD+ precursors may serve as an effective therapeutic strategy that limits mast cell-mediated anaphylactic responses.
Keywords: Sirt6, mast cell, anaphylaxis, NMN, NR
 Global reach, higher impact
Global reach, higher impact