Impact Factor
Theranostics 2022; 12(7):3057-3078. doi:10.7150/thno.72015 This issue Cite
Research Paper
hUC-MSC-mediated recovery of subacute spinal cord injury through enhancing the pivotal subunits β3 and γ2 of the GABAA receptor
1. Department of Cell Biology, School of Basic Medical Sciences, Southern Medical University, Guangzhou, 510515, China.
2. Department of Spine Surgery, The Third Affiliated Hospital of Sun Yat-sen University, Guangzhou, 510515, China.
3. Guangdong Provincial Center for Engineering and Technology Research of Minimally Invasive Spine Surgery, 510630, China.
4. Guangdong Provincial Center for Quality Control of Minimally Invasive Spine Surgery, 510630, China.
*These authors contributed equally to this work.
Abstract
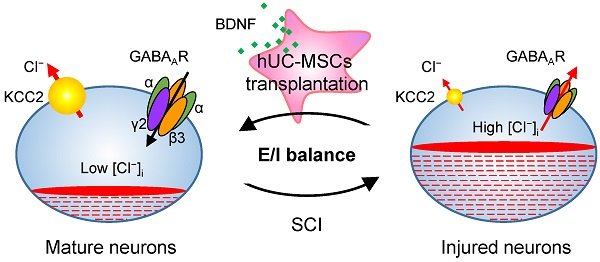
Rationale: Spinal cord injury (SCI) remains an incurable neurological disorder leading to permanent and profound neurologic deficits and disabilities. Human umbilical cord-derived mesenchymal stem cells (hUC-MSCs) are particularly appealing in SCI treatment to curtail damage, restore homeostasis and possible neural relay. However, the detailed mechanisms underlying hUC-MSC-mediated functional recovery of SCI have not been fully elucidated. The purpose of our current study is to identify novel therapeutic targets and depict the molecular mechanisms underlying the hUC-MSC-mediated recovery of subacute SCI.
Methods: Adult female rats suffering from subacute incomplete thoracic SCI were treated with intrathecal transplantation of hUC-MSCs. The beneficial effects of hUC-MSCs on SCI repair were evaluated by a series of behavioral analyses, motor evoked potentials (MEPs) recording of hindlimb and immunohistochemistry. We carried out extensive transcriptome comparative analyses of spinal cord tissues at the lesion site from the subacute phase of SCI (sub-SCI) either treated without (+PBS) or with hUC-MSCs (+MSC) at 0 (sub-SCI), 1, 2, and 4 weeks post-transplantation (wpt), as well as normal spinal cord segments of intact/sham rats (Intact). Adeno-associated virus (AAV)-mediated neuron-specific expression system was employed to functionally screen specific γ-aminobutyric acid type A receptor (GABAAR) subunits promoting the functional recovery of SCI in vivo. The mature cortical axon scrape assay and transplantation of genetically modified MSCs with either overexpression or knockdown of brain-derived neurotrophic factor (BDNF) were employed to demonstrate that hUC-MSCs ameliorated the reduction of GABAAR subunits in the injured spinal cord via BDNF secretion in vitro and in vivo, respectively.
Results: Comparative transcriptome analysis revealed the GABAergic synapse pathway is significantly enriched as a main target of hUC-MSC-activated genes in the injured spinal cord. Functional screening of the primary GABAAR subunits uncovered that Gabrb3 and Garbg2 harbored the motor and electrophysiological recovery-promoting competence. Moreover, targeting either of the two pivotal subunits β3 or γ2 in combination with/without the K+/Cl- cotransporter 2 (KCC2) reinforced the therapeutic effects. Mechanistically, BDNF secreted by hUC-MSCs contributed to the upregulation of GABAAR subunits (β3 & γ2) and KCC2 in the injured neurons.
Conclusions: Our study identifies a novel mode for hUC-MSC-mediated locomotor recovery of SCI through synergistic upregulation of GABAAR β3 and γ2 along with KCC2 by BDNF secretion, indicating the significance of restoring the excitation/inhibition balance in the injured neurons for the reestablishment of neuronal circuits. This study also provides a potential combinatorial approach by targeting the pivotal subunit β3 or γ2 and KCC2, opening up possibilities for efficacious drug design.
Keywords: spinal cord injury, human umbilical cord mesenchymal stem cells, transcriptome, γ-aminobutyric acid type A (GABAA) receptor, K+-Cl⁻ cotransporter 2
 Global reach, higher impact
Global reach, higher impact