Impact Factor
Theranostics 2022; 12(6):2928-2947. doi:10.7150/thno.69533 This issue Cite
Research Paper
AdMSC-derived exosomes alleviate acute lung injury via transferring mitochondrial component to improve homeostasis of alveolar macrophages
1. Key Laboratory of Inflammation and Immunoregulation, School of Medicine and Holistic Integrative Medicine, Nanjing University of Chinese Medicine, Nanjing City 210046, China
2. Department of Clinical Research, Friendship Plastic Surgery Hospital, Nanjing Medical University, Nanjing City 210029, China
3. School of Medicine and Holistic Integrative Medicine, Nanjing University of Chinese Medicine, Nanjing City 210046, China
4. The First School of Clinical Medicine, Affiliated Hospital of Nanjing University of Chinese Medicine, Nanjing City 210029, China
5. Affiliated Hospital of Xuzhou Medical University, Xuzhou Medical University, Xuzhou City 221004, China
6. Acupuncture and Tuina College, Nanjing University of Chinese Medicine, Nanjing City 210023, China
7. Shulan International Medical College, Zhejiang Shuren University, Hangzhou City 310022, China
Abstract
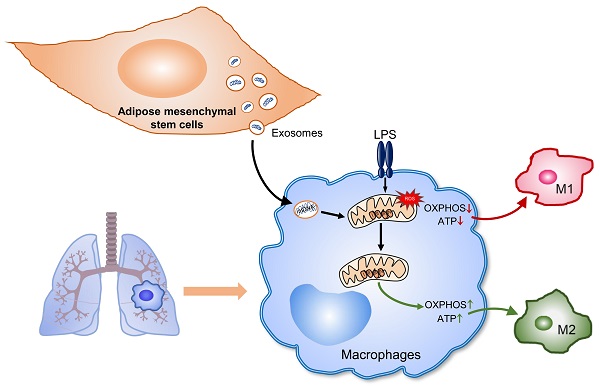
Rationale: Aberrant activation of macrophages with mitochondria dismiss was proved to be associated with pathogenesis of ALI (acute lung injury). Exosomes from adipose-derived mesenchymal stem cells (AdMSC-Exos) have been distinguished by their low immunogenicity, lack of tumorigenicity, and high clinical safety, but their role in treating ALI and the mechanism involved need to be defined. In this study, we sought to investigate whether the mitochondrial donation from AdMSC-Exos provides profound protection against LPS-induced ALI in mice, accompanied by improvement of macrophage mitochondrial function.
Methods: C57BL/6 mice were orotracheally instilled with LPS (1 mg/kg). AdMSC-Exos were administered via the tail vein 4 h after LPS inhalation. Flow cytometry, H&E, Quantitative Real-Time PCR, immunofluorescence (IF), confocal microscopy imaging was conducted to investigate lung tissue inflammation and macrophage mitochondrial function. And further observe the transfer of exosomes and the effect on mitochondrial function of MH-S cells through in vitro experiments.
Results: AdMSC-Exos can transfer the stem cell-derived mitochondria components to alveolar macrophages in a dose-dependent manner. Likely through complementing the damaged mitochondria, AdMSC-Exos exhibited the ability to elevate the level of mtDNA, mitochondrial membrane potential (MMP), OXPHOS activity and ATP generation, while reliving mROS stress in LPS-challenged macrophages. Restoring mitochondrial integrity via AdMSC-Exos treatment enabled macrophages shifting to anti-inflammatory phenotype, as featured with the down-regulation of IL-1β, TNF-α and iNOS secretion and increase in production of anti-inflammatory cytokines IL-10 and Arg-1. As we depleted alveolar macrophages using clodronate liposomes, the protective role for AdMSC-Exos was largely abrogated.
Conclusions: AdMSC-Exos can effectively donate mitochondria component improved macrophages mitochondrial integrity and oxidative phosphorylation level, leading to the resumption of metabolic and immune homeostasis of airway macrophages and mitigating lung inflammatory pathology.
Keywords: stem cells, exosomes, acute lung injury, alveolar macrophages, mitochondrial function
 Global reach, higher impact
Global reach, higher impact