13.3
Impact Factor
Theranostics 2022; 12(6):2687-2706. doi:10.7150/thno.67954 This issue Cite
Research Paper
Disease-associated microglial activation prevents photoreceptor degeneration by suppressing the accumulation of cell debris and neutrophils in degenerating rat retinas
1. Southwest Hospital/Southwest Eye Hospital, Third Military Medical University (Amy Medical University), Chongqing, 400038, P.R. China
2. Key Lab of Visual Damage and Regeneration & Restoration of Chongqing, Chongqing, 400038, P.R. China
3. No. 927 Hospital, Joint Logistics Support Force of Chinese PLA, Puer 665000, Yunnan, China
4. The General Hospital of Western Theater Command, Chengdu 610083, China
Abstract
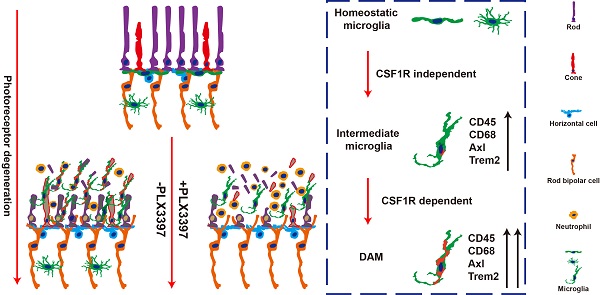
Retinitis pigmentosa initially presents as night blindness owing to defects in rods, and the secondary degeneration of cones ultimately leads to blindness. Previous studies have identified active roles of microglia in the pathogenesis of photoreceptor degeneration in RP. However, the contribution of microglia to photoreceptor degeneration remains controversial, partly due to limited knowledge of microglial phenotypes during RP.
Rationale: In this study, we investigated the pathways of microglial activation and its contribution to photoreceptor degeneration in RP.
Methods: A classic RP model, Royal College of Surgeons rat, was used to explore the process of microglial activation during the development of RP. An inhibitor of colony-stimulating factor 1 receptor (PLX3397) was fed to RCS rats for sustained ablation of microglia. Immunohistochemistry, flow cytometry, RT-qPCR, electroretinography and RNA-Seq were used to investigate the mechanisms by which activated microglia influenced photoreceptor degeneration.
Results: Microglia were gradually activated to disease-associated microglia in the photoreceptor layers of RCS rats. Sustained treatment with PLX3397 ablated most of the disease-associated microglia and aggravated photoreceptor degeneration, including the secondary degeneration of cones, by downregulating the expression of genes associated with photoreceptor function and components and exacerbating the impairment of photoreceptor cell function. Disease-associated microglial activation promoted microglia to engulf apoptotic photoreceptor cell debris and suppressed the increase of infiltrated neutrophils by increasing engulfment and inhibiting CXCL1 secretion by Müller cells, which provided a healthier microenvironment for photoreceptor survival.
Conclusions: Our data highlight a key role of disease-associated microglia activation in the suppression of rod and cone degeneration, which reduces secondary damage caused by the accumulation of dead cells and infiltrated neutrophils in the degenerating retina.
Keywords: microglia, disease-associated microglia, photoreceptor degeneration, neutrophil, photoreceptor
 Global reach, higher impact
Global reach, higher impact