13.3
Impact Factor
Theranostics 2022; 12(6):2519-2534. doi:10.7150/thno.67038 This issue Cite
Research Paper
Inhibitor screening using microarray identifies the high capacity of neutralizing antibodies to Spike variants in SARS-CoV-2 infection and vaccination
1. State Key Laboratory of Proteomics, Beijing Proteome Research Center, National Center for Protein Sciences-Beijing (PHOENIX Center), Beijing Institute of Lifeomics, Beijing, 102206, China.
2. Department of Clinical Laboratory, Beijing Ditan Hospital, Capital Medical University, Beijing, 100102, China.
3. Department of Research Ward, Beijing Ditan Hospital, Capital Medical University, Beijing, 100102, China.
4. State Key Laboratory of Infectious Disease Prevention and Control, National Institute for Communicable Disease Control and Prevention, Chinese Center for Disease Control and Prevention, Beijing, 102206, China
5. Joint Laboratory for Pathogen Identification of ICDC and Ditan Hospital, Beijing Ditan Hospital, Capital Medical University, Beijing, 100102, China
6. Beijing Key Laboratory of Monoclonal Antibody Research and Development, Sino Biological, Inc., Beijing, 100176, China
7. Gobond Testing Technology (Beijing) Co., Ltd., Beijing, 102629, China.
*These authors contributed equally to this work.
Abstract
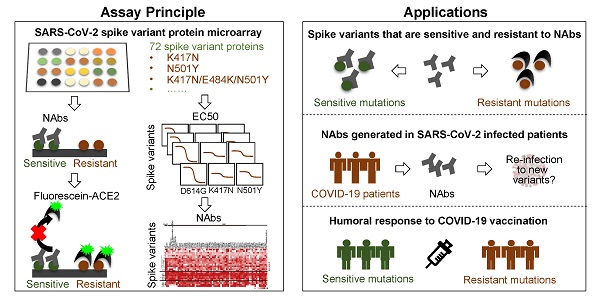
Rationale: Mutations of SARS-CoV-2, which is responsible for coronavirus disease 2019 (COVID-19), could impede drug development and reduce the efficacy of COVID-19 vaccines. Here, we developed a multiplexed Spike-ACE2 Inhibitor Screening (mSAIS) assay that can measure the neutralizing effect of antibodies across numerous variants of the coronavirus's Spike (S) protein simultaneously.
Methods: The SARS-CoV-2 spike variant protein microarrays were prepared by printing 72 S variants onto a chemically-modified glass slides. The neutralization potential of purified anti-S antibodies and serum from convalescent COVID-19 patients and vaccinees to S variants were assessed with the mSAIS assay.
Results: We identified new S mutations that are sensitive and resistant to neutralization. Serum from both infected and vaccinated groups with a high titer of neutralizing antibodies (NAbs) displayed a broader capacity to neutralize S variants than serum with low titer NAbs. These data were validated using serum from a large vaccinated cohort (n = 104) with a tiled S peptide microarray. In addition, similar results were obtained using a SARS-CoV-2 pseudovirus neutralization assay specific for wild-type S and five prevalent S variants (D614G, B.1.1.7, B.1.351, P.1, B.1.617.2), thus demonstrating that high antibody diversity is associated with high NAb titers.
Conclusions: Our results demonstrate the utility of the mSAIS platform in screening NAbs. Moreover, we show that heterogeneous antibody populations provide a more protective effect against S variants, which may help direct COVID-19 vaccine and drug development.
Keywords: SARS-CoV-2, microarray, neutralizing antibody, mutation, Spike
 Global reach, higher impact
Global reach, higher impact