13.3
Impact Factor
Theranostics 2022; 12(4):1621-1638. doi:10.7150/thno.64963 This issue Cite
Research Paper
Piezo1-mediated mechanosensation in bone marrow macrophages promotes vascular niche regeneration after irradiation injury
1. Laboratory of Radiation Biology, Laboratory Medicine Center, Department of Blood Transfusion, The Second Affiliated Hospital, Army Military Medical University, Chongqing 400037, China
2. 111 Project Laboratory of Biomechanics and Tissue Repair, College of Bioengineering, Chongqing University, Chongqing 400044, China
3. Neuro-oncology and Stem Cell Research Laboratory, CHOC Children's Research Institute, Children's Hospital of Orange County (CHOC); 1201 La Veta Ave., Orange, CA 92868-3874, USA
4. Department of Neurology, University of California - Irvine School of Medicine, 200 S. Manchester Ave. Ste. 206, Orange, CA 92868, USA
Abstract
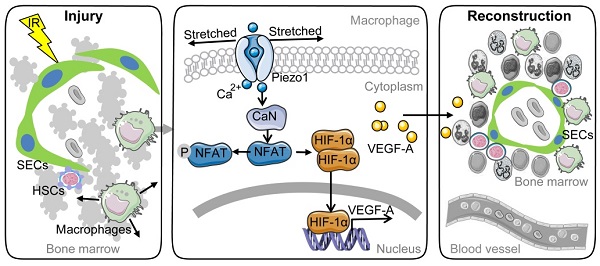
Background: Irradiation disrupts the vascular niche where hematopoietic stem cells (HSCs) reside, causing delayed hematopoietic reconstruction. The subsequent recovery of sinusoidal vessels is key to vascular niche regeneration and a prerequisite for hematopoietic reconstruction. We hypothesize that resident bone marrow macrophages (BM-Mφs) are responsible for repairing the HSC niche upon irradiation injury.
Methods: We examined the survival and activation of BM-Mφs in C57BL/6 mice upon total body irradiation. After BM-Mφ depletion via injected clodronate-containing liposomes and irradiation injury, hematopoietic reconstruction and sinusoidal vascular regeneration were assessed with immunofluorescence and flow cytometry. Then enzyme-linked immunosorbent assay (ELISA) and flow cytometry were performed to analyze the contribution of VEGF-A released by BM-Mφs to the vascular restructuring of the HSC niche. VEGF-A-mediated signal transduction was assessed with transcriptome sequencing, flow cytometry, and pharmacology (agonists and antagonists) to determine the molecular mechanisms of Piezo1-mediated responses to structural changes in the HSC niche.
Results: The depletion of BM-Mφs aggravated the post-irradiation injury, delaying the recovery of sinusoidal endothelial cells and HSCs. A fraction of the BM-Mφ population persisted after irradiation, with residual BM-Mφ exhibiting an activated M2-like phenotype. The expression of VEGF-A, which is essential for sinusoidal regeneration, was upregulated in BM-Mφs post-irradiation, especially CD206+ BM-Mφs. The expression of mechanosensory ion channel Piezo1, a response to mechanical environmental changes induced by bone marrow ablation, was upregulated in BM-Mφs, especially CD206+ BM-Mφs. Piezo1 upregulation was mediated by the effects of irradiation, the activation of Piezo1 itself, and the M2-like polarization induced by the phagocytosis of apoptotic cells. Piezo1 activation was associated with increased expression of VEGF-A and increased accumulation of NFATC1, NFATC2, and HIF-1α. The Piezo1-mediated upregulation in VEGF-A was suppressed by inhibiting the calcineurin/NFAT/HIF-1α signaling pathway.
Conclusion: These findings reveal that BM-Mφs play a critical role in promoting vascular niche regeneration by sensing and responding to structural changes after irradiation injury, offering a potential target for therapeutic efforts to enhance hematopoietic reconstruction.
Keywords: irradiation, Piezo1, macrophages, sinusoidal regeneration, hematopoietic reconstitution
 Global reach, higher impact
Global reach, higher impact