13.3
Impact Factor
Theranostics 2022; 12(4):1589-1606. doi:10.7150/thno.69054 This issue Cite
Research Paper
PPAR-γ integrates obesity and adipocyte clock through epigenetic regulation of Bmal1
1. Institute of molecular rhythm and metabolism, Guangzhou University of Chinese Medicine, Guangzhou 510405, China.
2. College of Pharmacy, Jinan University, Guangzhou 510632, China.
3. The First Affiliated Hospital of Jinan University, Guangzhou 510632, China.
# These authors contribute equally to this work.
Abstract
While growing evidence suggests that circadian clock and obesity are intertwined, the underlying mechanism is poorly understood. Here, we investigate how circadian clock is linked to obesity.
Methods: Metabolomics profiling of WAT (white adipose tissue) samples was performed to identify the metabolites altered in obese model. mRNA levels were analyzed by qPCR assays. Proteins were detected by immunoblotting, immunofluorescence and ELISA. ChIP and luciferase reporter assays were used to investigate epigenetic and transcriptional regulation.
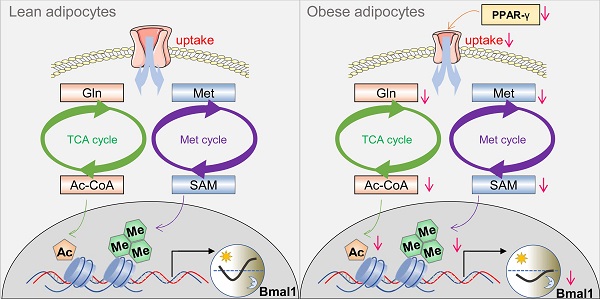
Results: Obesity causes perturbance of circadian clock in WAT in mice and humans, particularly, BMAL1 is markedly reduced. Metabolomic analysis reveals reduced glutamine and methionine in obese WAT. Glutamine metabolism contributes to production of acetyl-CoA, whereas methionine metabolism generates S-adenosyl methionine (SAM). Acetyl-CoA and SAM are the substrates for histone acetylation and methylation, respectively. Reduced glutamine and methionine in obese WAT are associated with decreased H3K27ac and H3K4me3 at Bmal1 promoter. Consistently, glutamine or methionine administration in vitro and in vivo increases H3K27ac or H3K4me3, promoting Bmal1 transcription and expression. A screen of transport and metabolic genes identifies downregulation of the uptake transporter SLC1A5 as a cause of reduced glutamine or methionine in obese WAT. Moreover, we observe impaired expression of PPAR-γ in obese WAT. PPAR-γ trans-activates Slc1a5 via direct binding to a response element in promoter.
Conclusion: Impaired PPAR-γ in obesity provokes downregulation of SLC1A5 and reductions in adipocyte uptake of glutamine and methionine (two epigenetic modulators), leading to disruption of Bmal1. Therefore, PPAR-γ integrates obesity and adipocyte clock, promoting a vicious cycle between circadian disruption and obesity development.
Keywords: PPAR-γ, obesity, circadian clock, BMAL1, SLC1A5
 Global reach, higher impact
Global reach, higher impact