13.3
Impact Factor
Theranostics 2022; 12(2):859-874. doi:10.7150/thno.66274 This issue Cite
Research Paper
Extracellular ATP is increased by release of ATP-loaded microparticles triggered by nutrient deprivation
1. Department of Medical Sciences, University of Ferrara, Ferrara, Italy.
2. Department of Inorganic, Organic Chemistry and Biochemistry, Universidad de Castilla-La Mancha, Regional Center of Biomedical Research, Ciudad Real, Spain.
3. Center of Electronic Microscopy, University of Ferrara, Ferrara, Italy.
4. Biosciences Laboratory, Istituto Scientifico Romagnolo per lo Studio e la Cura dei Tumori (IRST), IRCCS, Meldola, Italy.
Abstract
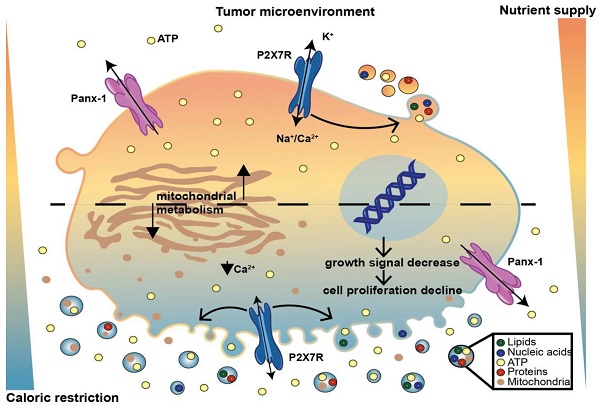
Rationale: Caloric restriction improves the efficacy of anti-cancer therapy. This effect is largely dependent on the increase of the extracellular ATP concentration in the tumor microenvironment (TME). Pathways for ATP release triggered by nutrient deprivation are largely unknown.
Methods: The extracellular ATP (eATP) concentration was in vivo measured in the tumor microenvironment of B16F10-inoculated C57Bl/6 mice with the pmeLuc probe. Alternatively, the pmeLuc-TG-mouse was used. Caloric restriction was in vivo induced with hydroxycitrate (HC). B16F10 melanoma cells or CT26 colon carcinoma cells were in vitro exposed to serum starvation to mimic nutrient deprivation. Energy metabolism was monitored by Seahorse. Microparticle release was measured by ultracentrifugation and by Nanosight.
Results: Nutrient deprivation increases eATP release despite the dramatic inhibition of intracellular energy synthesis. Under these conditions oxidative phosphorylation was dramatically impaired, mitochondria fragmented and glycolysis and lactic acid release were enhanced. Nutrient deprivation stimulated a P2X7-dependent release of ATP-loaded, mitochondria-containing, microparticles as well as of naked mitochondria.
Conclusions: Nutrient deprivation promotes a striking accumulation of eATP paralleled by a large release of ATP-laden microparticles and of naked mitochondria. This is likely to be a main mechanism driving the accumulation of eATP into the TME.
Keywords: nutrient deprivation, extracellular ATP, microparticles, tumor microenvironment, P2X7.
 Global reach, higher impact
Global reach, higher impact