13.3
Impact Factor
Theranostics 2022; 12(2):842-858. doi:10.7150/thno.66575 This issue Cite
Research Paper
FcγRIIB potentiates differentiation of myeloid-derived suppressor cells to mediate tumor immunoescape
1. Department of Medical Oncology, Chongqing University Cancer Hospital, Chongqing 400030, China.
2. Chongqing Key Laboratory for Intelligent Oncology in Breast Cancer, Chongqing University Cancer Hospital, Chongqing 400030, China.
3. Clinical Medicine Research Center, Xinqiao Hospital, Army Medical University, Chongqing 400037, China.
4. Department of Radiotherapy, Chongqing University Cancer Hospital, Chongqing 400030, China.
Abstract
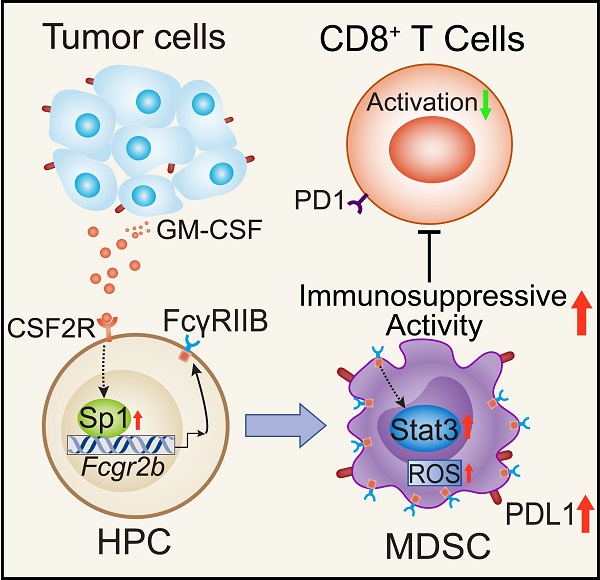
Background: FcγRIIB, the sole inhibitory receptor of the Fc gamma receptor family, plays pivotal roles in innate and adaptive immune responses. However, the expression and function of FcγRIIB in myeloid-derived suppressor cells (MDSCs) remains unknown. This study aimed to investigate whether and how FcγRIIB regulates the immunosuppressive activity of MDSCs during cancer development.
Methods: The MC38 and B16-F10 tumor-bearing mouse models were established to investigate the role of FcγRIIB during tumor progression. FcγRIIB-deficient mice, adoptive cell transfer, mRNA-sequencing and flow cytometry analysis were used to assess the role of FcγRIIB on immunosuppressive activity and differentiation of MDSCs.
Results: Here we show that FcγRIIB was upregulated in tumor-infiltrated MDSCs. FcγRIIB-deficient mice showed decreased accumulation of MDSCs in the tumor microenvironment (TME) compared with wild-type mice. FcγRIIB was required for the differentiation and immunosuppressive activity of MDSCs. Mechanistically, tumor cell-derived granulocyte-macrophage colony stimulating factor (GM-CSF) increased the expression of FcγRIIB on hematopoietic progenitor cells (HPCs) by activating specificity protein 1 (Sp1), subsequently FcγRIIB promoted the generation of MDSCs from HPCs via Stat3 signaling. Furthermore, blockade of Sp1 dampened MDSC differentiation and infiltration in the TME and enhanced the anti-tumor therapeutic efficacy of gemcitabine.
Conclusion: These results uncover an unrecognized regulatory role of the FcγRIIB in abnormal differentiation of MDSCs during cancer development and suggest a potential therapeutic target for anti-tumor therapy.
Keywords: myeloid-derived suppressor cells, Fc gamma receptor IIB, tumor microenvironment, granulocyte-macrophage colony stimulating factor, immunosuppression, anti-tumor therapy, Sp1 signaling
 Global reach, higher impact
Global reach, higher impact